Photopigment quenching is Ca2+ dependent and controls response duration in salamander L-cone photoreceptors
- PMID: 20231373
- PMCID: PMC2847922
- DOI: 10.1085/jgp.200910394
Photopigment quenching is Ca2+ dependent and controls response duration in salamander L-cone photoreceptors
Abstract
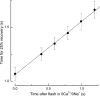
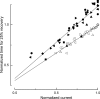
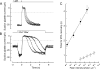
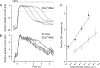
The time scale of the photoresponse in photoreceptor cells is set by the slowest of the steps that quench the light-induced activity of the phototransduction cascade. In vertebrate photoreceptor cells, this rate-limiting reaction is thought to be either shutoff of catalytic activity in the photopigment or shutoff of the pigment's effector, the transducin-GTP-phosphodiesterase complex. In suction pipette recordings from isolated salamander L-cones, we found that preventing changes in internal [Ca(2+)] delayed the recovery of the light response and prolonged the dominant time constant for recovery. Evidence that the Ca(2+)-sensitive step involved the pigment itself was provided by the observation that removal of Cl(-) from the pigment's anion-binding site accelerated the dominant time constant for response recovery. Collectively, these observations indicate that in L-cones, unlike amphibian rods where the dominant time constant is insensitive to [Ca(2+)], pigment quenching rate limits recovery and provides an additional mechanism for modulating the cone response during light adaptation.
Figures



Similar articles
-
Origin and control of the dominant time constant of salamander cone photoreceptors.J Gen Physiol. 2012 Aug;140(2):219-33. doi: 10.1085/jgp.201110762. Epub 2012 Jul 16. J Gen Physiol. 2012. PMID: 22802362 Free PMC article.
-
Light-dependent changes in outer segment free-Ca2+ concentration in salamander cone photoreceptors.J Gen Physiol. 1999 Feb;113(2):267-77. doi: 10.1085/jgp.113.2.267. J Gen Physiol. 1999. PMID: 9925824 Free PMC article.
-
Temperature dependence of dark-adapted sensitivity and light-adaptation in photoreceptors with A1 visual pigments: a comparison of frog L-cones and rods.Vision Res. 2009 Jul;49(14):1717-28. doi: 10.1016/j.visres.2009.03.024. Epub 2009 Apr 5. Vision Res. 2009. PMID: 19348836
-
Rod and cone photoreceptors: molecular basis of the difference in their physiology.Comp Biochem Physiol A Mol Integr Physiol. 2008 Aug;150(4):369-77. doi: 10.1016/j.cbpa.2008.04.600. Epub 2008 Apr 26. Comp Biochem Physiol A Mol Integr Physiol. 2008. PMID: 18514002 Review.
-
Molecular mechanisms characterizing cone photoresponses.Photochem Photobiol. 2007 Jan-Feb;83(1):19-26. doi: 10.1562/2006-02-28-IR-823. Photochem Photobiol. 2007. PMID: 16706600 Review.
Cited by
-
Speed, adaptation, and stability of the response to light in cone photoreceptors: the functional role of Ca-dependent modulation of ligand sensitivity in cGMP-gated ion channels.J Gen Physiol. 2012 Jan;139(1):31-56. doi: 10.1085/jgp.201110654. J Gen Physiol. 2012. PMID: 22200947 Free PMC article.
-
Photoreceptor signaling: supporting vision across a wide range of light intensities.J Biol Chem. 2012 Jan 13;287(3):1620-6. doi: 10.1074/jbc.R111.305243. Epub 2011 Nov 10. J Biol Chem. 2012. PMID: 22074925 Free PMC article. Review.
-
Multi-scale, numerical modeling of spatio-temporal signaling in cone phototransduction.PLoS One. 2019 Jul 25;14(7):e0219848. doi: 10.1371/journal.pone.0219848. eCollection 2019. PLoS One. 2019. PMID: 31344066 Free PMC article.
-
Activation and quenching of the phototransduction cascade in retinal cones as inferred from electrophysiology and mathematical modeling.Mol Vis. 2015 Mar 7;21:244-63. eCollection 2015. Mol Vis. 2015. PMID: 25866462 Free PMC article.
-
Circadian regulation of vertebrate cone photoreceptor function.Elife. 2021 Sep 22;10:e68903. doi: 10.7554/eLife.68903. Elife. 2021. PMID: 34550876 Free PMC article.
References
-
- Bownds D., Dawes J., Miller J., Stahlman M. 1972. Phosphorylation of frog photoreceptor membranes induced by light. Nat. New Biol. 237:125–127 - PubMed

