Use of the Cre-lox recombination system to investigate the lp54 gene requirement in the infectious cycle of Borrelia burgdorferi
- PMID: 20231410
- PMCID: PMC2876536
- DOI: 10.1128/IAI.01059-09
Use of the Cre-lox recombination system to investigate the lp54 gene requirement in the infectious cycle of Borrelia burgdorferi
Abstract
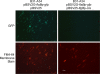
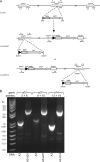
Borrelia burgdorferi, the causative agent of Lyme disease, has a complex genome consisting of a linear chromosome and up to 21 linear and circular plasmids. These plasmids encode numerous proteins critical to the spirochete's infectious cycle and many hypothetical proteins whose functions and requirements are unknown. The conserved linear plasmid lp54 encodes several proteins important for survival in the mouse-tick infectious cycle, but the majority of the proteins are of unknown function and lack homologs outside the borreliae. In this study we adapted the Cre-lox recombination system to create large deletions in the B. burgdorferi genome. Using Cre-lox, we systematically investigated the contribution of 14 adjacent genes on the left arm of lp54 to the overall infectivity of B. burgdorferi. The deletion of the region of lp54 encompassing bba07 to bba14 had no significant effect on the infectious cycle of B. burgdorferi. The deletion of bba01 to bba07 resulted in a slight growth defect but did not significantly affect the ability of B. burgdorferi to complete the infectious cycle. This study demonstrated the utility of the Cre-lox system to efficiently explore gene requirements in B. burgdorferi and surprisingly revealed that a large number of the highly conserved proteins encoded on lp54 are not required to complete the infectious cycle.
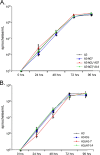
Figures

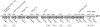

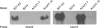
References
-
- Beaurepaire, C., and G. Chaconas. 2005. Mapping of essential replication functions of the linear plasmid lp17 of Borrelia burgdorferi by targeted deletion walking. Mol. Microbiol. 57:132-142. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

