Fluorescence polarization/anisotropy in diagnostics and imaging
- PMID: 20232898
- PMCID: PMC2868933
- DOI: 10.1021/cr900267p
Fluorescence polarization/anisotropy in diagnostics and imaging
Figures


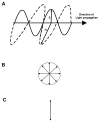
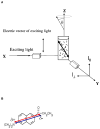

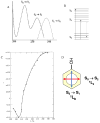
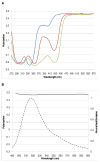


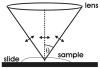
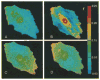

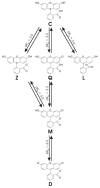
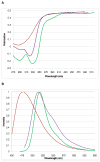
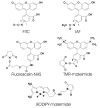
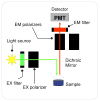
References
-
- Jameson DM. In: New Trends in Fluorescence Spectroscopy. Valeur B, Brochon JC, editors. Springer; Heidelberg: 2001.
-
- Malus EL. Nouveau Bull de la Societé Philomatique. 1809;1:266.
-
- Brewster D. Philos Trans R Soc London. 1830;2:387.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

