STAT2 contributes to promotion of colorectal and skin carcinogenesis
- PMID: 20233899
- PMCID: PMC2851485
- DOI: 10.1158/1940-6207.CAPR-09-0105
STAT2 contributes to promotion of colorectal and skin carcinogenesis
Abstract
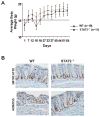
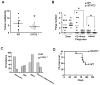
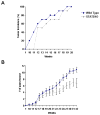
Signal transducer and activator of transcription 2 (STAT2) is an essential transcription factor in the type I IFN (IFN-alpha/beta) signal transduction pathway and known for its role in mediating antiviral immunity and cell growth inhibition. Unlike other members of the STAT family, IFNs are the only cytokines known to date that can activate STAT2. Given the inflammatory and antiproliferative dual nature of IFNs, we hypothesized that STAT2 prevents inflammation-induced colorectal and skin carcinogenesis by altering the inflammatory immune response. Contrary to our hypothesis, deletion of STAT2 inhibited azoxymethane/dextran sodium sulfate-induced colorectal carcinogenesis as measured by prolonged survival, lower adenoma incidence, smaller polyps, and less chronic inflammation. STAT2 deficiency also inhibited 7,12-dimethylbenz(a)anthracene/12-O-tetradecanoylphorbol-13-acetate-induced skin carcinogenesis as indicated by reduced papilloma multiplicity. A potential mechanism by which STAT2 promotes carcinogenesis is through activation of proinflammatory mediators. Deletion of STAT2 decreased azoxymethane/dextran sodium sulfate-induced expression and release of proinflammatory mediators, such as interleukin-6 and CCL2, and decreased interleukin-6 release from skin carcinoma cells, which then decreased STAT3 activation. Our findings identify STAT2 as a novel contributor to colorectal and skin carcinogenesis that may act to increase the gene expression and secretion of proinflammatory mediators, which in turn activate the oncogenic STAT3 signaling pathway.
(c) 2010 AACR.
Figures



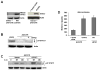
Similar articles
-
Dominant negative signal transducer and activator of transcription 2 (STAT2) protein: stable expression blocks interferon alpha action in skin squamous cell carcinoma cells.Mol Cancer Ther. 2003 May;2(5):453-9. Mol Cancer Ther. 2003. PMID: 12748307
-
Stage-specific disruption of Stat3 demonstrates a direct requirement during both the initiation and promotion stages of mouse skin tumorigenesis.Carcinogenesis. 2008 Jun;29(6):1108-14. doi: 10.1093/carcin/bgn061. Epub 2008 May 2. Carcinogenesis. 2008. PMID: 18453544 Free PMC article.
-
Stat2 stability regulation: an intersection between immunity and carcinogenesis.Exp Mol Med. 2020 Sep;52(9):1526-1536. doi: 10.1038/s12276-020-00506-6. Epub 2020 Sep 25. Exp Mol Med. 2020. PMID: 32973222 Free PMC article. Review.
-
PDCD4 Deficiency Aggravated Colitis and Colitis-associated Colorectal Cancer Via Promoting IL-6/STAT3 Pathway in Mice.Inflamm Bowel Dis. 2016 May;22(5):1107-18. doi: 10.1097/MIB.0000000000000729. Inflamm Bowel Dis. 2016. PMID: 26891257
-
The role of signal transducer and activator of transcription-2 in the interferon response.J Interferon Cytokine Res. 2012 Mar;32(3):103-10. doi: 10.1089/jir.2011.0099. Epub 2012 Jan 26. J Interferon Cytokine Res. 2012. PMID: 22280068 Free PMC article. Review.
Cited by
-
Nanoparticles Targeting STATs in Cancer Therapy.Cells. 2019 Sep 27;8(10):1158. doi: 10.3390/cells8101158. Cells. 2019. PMID: 31569687 Free PMC article. Review.
-
FBXW7-mediated stability regulation of signal transducer and activator of transcription 2 in melanoma formation.Proc Natl Acad Sci U S A. 2020 Jan 7;117(1):584-594. doi: 10.1073/pnas.1909879116. Epub 2019 Dec 16. Proc Natl Acad Sci U S A. 2020. PMID: 31843895 Free PMC article.
-
Multi-omics analysis for potential inflammation-related genes involved in tumour immune evasion via extended application of epigenetic data.Open Biol. 2022 Aug;12(8):210375. doi: 10.1098/rsob.210375. Epub 2022 Aug 10. Open Biol. 2022. PMID: 35946310 Free PMC article.
-
STAT3 drives the malignant progression of low-grade gliomas through modulating the expression of STAT1, FOXO1, and MYC.Front Mol Biosci. 2024 Jun 14;11:1419072. doi: 10.3389/fmolb.2024.1419072. eCollection 2024. Front Mol Biosci. 2024. PMID: 38948079 Free PMC article.
-
Filaggrin genotype determines functional and molecular alterations in skin of patients with atopic dermatitis and ichthyosis vulgaris.PLoS One. 2011;6(12):e28254. doi: 10.1371/journal.pone.0028254. Epub 2011 Dec 2. PLoS One. 2011. PMID: 22164253 Free PMC article.
References
-
- Balkwill F, Charles KA, Mantovani A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer cell. 2005;7:211–7. - PubMed
-
- Ekbom A. Risk of cancer in ulcerative colitis. J Gastrointest Surg. 1998;2:312–3. - PubMed
-
- Xiao H, Gulen MF, Qin J, et al. The Toll-interleukin-1 receptor member SIGIRR regulates colonic epithelial homeostasis, inflammation, and tumorigenesis. Immunity. 2007;26:461–75. - PubMed
-
- Greten FR, Eckmann L, Greten TF, et al. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell. 2004;118:285–96. - PubMed
-
- Pikarsky E, Porat RM, Stein I, et al. NF-kappaB functions as a tumour promoter in inflammation-associated cancer. Nature. 2004;431:461–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

