Systemic therapy of disseminated myeloma in passively immunized mice using measles virus-infected cell carriers
- PMID: 20234340
- PMCID: PMC2889728
- DOI: 10.1038/mt.2010.43
Systemic therapy of disseminated myeloma in passively immunized mice using measles virus-infected cell carriers
Abstract
Multiple myeloma (MM) is bone marrow plasma cell malignancy. A clinical trial utilizing intravenous administration of oncolytic measles virus (MV) encoding the human sodium-iodide symporter (MV-NIS) is ongoing in myeloma patients. However, intravenously administered MV-NIS is rapidly neutralized by antiviral antibodies. Because myeloma cell lines retain bone marrow tropism, they may be ideal as carriers for delivery of MV-NIS to myeloma deposits. A disseminated human myeloma (KAS 6/1) model was established. Biodistribution of MM1, a myeloma cell line, was determined after intravenous infusion. MM1 cells were found in the spine, femurs, and mandibles of tumor-bearing mice. Lethally irradiated MM1 cells remained susceptible to measles infection and transferred MV to KAS 6/1 cells in the presence of measles immune sera. Mice-bearing disseminated myeloma and passively immunized with measles immune serum were given MV-NIS or lethally irradiated MV-NIS-infected MM1 carriers. The antitumor activity of MV-NIS was evident only in measles naive mice and not in passively immunized mice. In contrast, survivals of both measles naive and immune mice were extended using MV-NIS-infected MM1 cell carriers. Hence, we demonstrate for the first time that systemically administered cells can serve as MV carriers and prolonged survival of mice with pre-existing antimeasles antibodies.
Figures
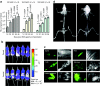
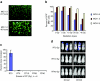
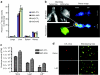
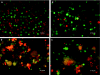


Similar articles
-
Evaluation of T cells as carriers for systemic measles virotherapy in the presence of antiviral antibodies.Gene Ther. 2007 Feb;14(4):324-33. doi: 10.1038/sj.gt.3302880. Epub 2006 Oct 19. Gene Ther. 2007. PMID: 17051248
-
Image-guided radiovirotherapy for multiple myeloma using a recombinant measles virus expressing the thyroidal sodium iodide symporter.Blood. 2004 Mar 1;103(5):1641-6. doi: 10.1182/blood-2003-07-2233. Epub 2003 Nov 6. Blood. 2004. PMID: 14604966
-
Mesenchymal stem cell carriers protect oncolytic measles viruses from antibody neutralization in an orthotopic ovarian cancer therapy model.Clin Cancer Res. 2009 Dec 1;15(23):7246-55. doi: 10.1158/1078-0432.CCR-09-1292. Epub 2009 Nov 24. Clin Cancer Res. 2009. PMID: 19934299 Free PMC article.
-
Cell carriers to deliver oncolytic viruses to sites of myeloma tumor growth.Gene Ther. 2008 May;15(10):797-806. doi: 10.1038/gt.2008.45. Epub 2008 Mar 20. Gene Ther. 2008. PMID: 18356812 Review.
-
Measles to the Rescue: A Review of Oncolytic Measles Virus.Viruses. 2016 Oct 22;8(10):294. doi: 10.3390/v8100294. Viruses. 2016. PMID: 27782084 Free PMC article. Review.
Cited by
-
Molecular imaging of oncolytic viral therapy.Mol Ther Oncolytics. 2015 Feb 4;1:14007. doi: 10.1038/mto.2014.7. eCollection 2015. Mol Ther Oncolytics. 2015. PMID: 27119098 Free PMC article. Review.
-
Oncolytic Viruses for Multiple Myeloma Therapy.Cancers (Basel). 2018 Jun 14;10(6):198. doi: 10.3390/cancers10060198. Cancers (Basel). 2018. PMID: 29903988 Free PMC article. Review.
-
Antitumor Virotherapy by Attenuated Measles Virus (MV).Biology (Basel). 2013 Mar 28;2(2):587-602. doi: 10.3390/biology2020587. Biology (Basel). 2013. PMID: 24832799 Free PMC article.
-
Measles Virus as an Oncolytic Immunotherapy.Cancers (Basel). 2021 Feb 1;13(3):544. doi: 10.3390/cancers13030544. Cancers (Basel). 2021. PMID: 33535479 Free PMC article. Review.
-
Cell Carriers for Oncolytic Virus Delivery: Prospects for Systemic Administration.Cancers (Basel). 2025 Jul 10;17(14):2296. doi: 10.3390/cancers17142296. Cancers (Basel). 2025. PMID: 40723180 Free PMC article. Review.
References
-
- Rajkumar SV., and , Kyle RA. Multiple myeloma: diagnosis and treatment. Mayo Clin Proc. 2005;80:1371–1382. - PubMed
-
- Dingli D., and , Rajkumar SV. Emerging therapies for multiple myeloma. Oncology (Williston Park, NY) 2009;23:407–415. - PubMed
-
- Parato KA, Lichty BD., and , Bell JC. Diplomatic immunity: turning a foe into an ally. Curr Opin Mol Ther. 2009;11:13–21. - PubMed
-
- Nguyen TL, Tumilasci VF, Singhroy D, Arguello M., and , Hiscott J. The emergence of combinatorial strategies in the development of RNA oncolytic virus therapies. Cell Microbiol. 2009;11:889–897. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

