Derivation of functional smooth muscle cells from multipotent human hair follicle mesenchymal stem cells
- PMID: 20236033
- PMCID: PMC2947431
- DOI: 10.1089/ten.TEA.2009.0833
Derivation of functional smooth muscle cells from multipotent human hair follicle mesenchymal stem cells
Abstract
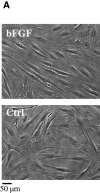
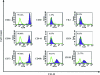
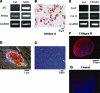
We investigated the potential of human hair follicle cells for multilineage differentiation and as a source of functional smooth muscle cells (SMCs). We report that human hair follicle stem cells (HFCs) isolated from individual follicles expressed surface markers that are characteristic of mesenchymal stem cells such as CD44, CD49b, CD73, CD90, and CD105 but lacked hematopoietic markers CD45 and CD34. In addition, HFCs differentiated toward adipocytes, chondrocytes, osteoblasts, or SMCs in the appropriate induction medium. Treatment with basic fibroblast growth factor increased proliferation and prevented myogenic differentiation, suggesting that basic fibroblast growth factor can be used to expand the population of undifferentiated HFCs to the large numbers needed for therapeutic applications. SMCs were isolated from HFCs using tissue-specific promoters and flow cytometry sorting. Cylindrical vascular constructs engineered with HF-SMCs showed remarkable contractility in response to receptor and nonreceptor agonists such KCl, endothelin-1, and the thromboxane mimetic, U46619, as well as superior mechanical properties compared to their counterparts with human vascular SMCs. Our results suggest that HF is a rich source of mesenchymal stem cells with great potential for myogenic differentiation providing functional SMCs for tissue regeneration and cell therapies.
Figures





References
-
- Alonso L. Fuchs E. The hair cycle. J Cell Sci. 2006;119:391. - PubMed
-
- Cotsarelis G. Sun T.T. Lavker R.M. Label-retaining cells reside in the bulge area of pilosebaceous unit: implications for follicular stem cells, hair cycle, and skin carcinogenesis. Cell. 1990;61:1329. - PubMed
-
- Morris R.J. Potten C.S. Highly persistent label-retaining cells in the hair follicles of mice and their fate following induction of anagen. J Invest Dermatol. 1999;112:470. - PubMed
-
- Morris R.J. Liu Y. Marles L. Yang Z. Trempus C. Li S. Lin J.S. Sawicki J.A. Cotsarelis G. Capturing and profiling adult hair follicle stem cells. Nat Biotechnol. 2004;22:411. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

