Regulation of Syk kinase and FcRbeta expression in human basophils during treatment with omalizumab
- PMID: 20236696
- PMCID: PMC2850964
- DOI: 10.1016/j.jaci.2009.12.996
Regulation of Syk kinase and FcRbeta expression in human basophils during treatment with omalizumab
Abstract
Background: In human basophils from different subjects, maximum IgE-mediated histamine release and the level of Syk protein expression correlate well. Recent studies suggest that in some patients treated with omalizumab, the response to stimulation with anti-IgE antibody increases. In unrelated studies there is also evidence that the composition of FcepsilonRI in basophils differs among subjects. This observation raised the possibility that the stoichiometry of FcRbeta/FcepsilonRIalpha is not fixed to a 1:1 ratio and might be modifiable during changes in the basophil's environment.
Objective: We sought to determine whether treatment with omalizumab results in increases in Syk expression and anti-IgE-mediated histamine release and disproportionately alters the relative presence of FcRbeta and FcepsilonRIalpha.
Method: Syk, FcepsilonRIalpha, and FcRbeta expression was monitored during the treatment of subjects with omalizumab.
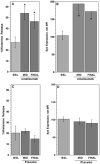
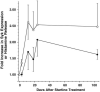
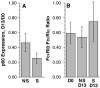
Results: Treatment with omalizumab reduced histamine release from peripheral blood leukocytes stimulated with cat allergen in vitro, but histamine release stimulated with anti-IgE antibody increased 2-fold. Expression of Syk increased 1.86-fold. There was no change in the expression of c-Cbl, a signaling element that is sensitive to the presence of IL-3, and no increase in response to formyl-met-leu-phe (tripeptide), a response that also increases in the presence of IL-3. There was a 60% decrease in the FcRbeta/FcepsilonRIalpha ratio in patients treated with omalizumab.
Conclusions: In the context of previous studies, these studies provide support for a proposal that Syk expression is modulated in vivo through an IgE-dependent mechanism and that the ratio of FcepsilonRI alpha and beta subunits in basophils is influenced by factors extrinsic to the cell.
Copyright (c) 2010 American Academy of Allergy, Asthma & Immunology. Published by Mosby, Inc. All rights reserved.
Figures
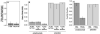
Similar articles
-
Omalizumab increases the intrinsic sensitivity of human basophils to IgE-mediated stimulation.J Allergy Clin Immunol. 2013 Oct;132(4):906-11.e1-4. doi: 10.1016/j.jaci.2013.04.056. Epub 2013 Jun 20. J Allergy Clin Immunol. 2013. PMID: 23791510 Free PMC article. Clinical Trial.
-
Response of peripheral blood basophils in subjects with chronic spontaneous urticaria during treatment with omalizumab.J Allergy Clin Immunol. 2021 Jun;147(6):2295-2304.e12. doi: 10.1016/j.jaci.2021.02.039. Epub 2021 Mar 12. J Allergy Clin Immunol. 2021. PMID: 33716077
-
Reduced FcepsilonRI-mediated release of asthma-promoting cytokines and chemokines from human basophils during omalizumab therapy.Int Arch Allergy Immunol. 2010;151(4):275-84. doi: 10.1159/000250436. Epub 2009 Oct 22. Int Arch Allergy Immunol. 2010. PMID: 19844128 Free PMC article.
-
Pathogenic intracellular and autoimmune mechanisms in urticaria and angioedema.Clin Rev Allergy Immunol. 2013 Aug;45(1):47-62. doi: 10.1007/s12016-012-8326-y. Clin Rev Allergy Immunol. 2013. PMID: 22674016 Review.
-
IgE antibody-specific activity in human allergic disease.Immunol Res. 2010 Jul;47(1-3):273-84. doi: 10.1007/s12026-009-8160-3. Immunol Res. 2010. PMID: 20066506 Review.
Cited by
-
Structural basis of omalizumab therapy and omalizumab-mediated IgE exchange.Nat Commun. 2016 May 19;7:11610. doi: 10.1038/ncomms11610. Nat Commun. 2016. PMID: 27194387 Free PMC article.
-
IL-3 but not monomeric IgE regulates FcεRI levels and cell survival in primary human basophils.Cell Death Dis. 2018 May 1;9(5):510. doi: 10.1038/s41419-018-0526-9. Cell Death Dis. 2018. PMID: 29724998 Free PMC article.
-
The relationship between released soluble FceRI-alpha and its cell surface density on human basophils.PLoS One. 2021 Jan 22;16(1):e0245942. doi: 10.1371/journal.pone.0245942. eCollection 2021. PLoS One. 2021. PMID: 33481953 Free PMC article.
-
Omalizumab and unmet needs in severe asthma and allergic comorbidities in Japanese children.Asia Pac Allergy. 2019 Jan 22;9(1):e7. doi: 10.5415/apallergy.2019.9.e7. eCollection 2019 Jan. Asia Pac Allergy. 2019. PMID: 30740355 Free PMC article. Review.
-
Stability of Syk protein and mRNA in human peripheral blood basophils.J Leukoc Biol. 2016 Sep;100(3):535-43. doi: 10.1189/jlb.2A0815-356R. Epub 2016 Mar 15. J Leukoc Biol. 2016. PMID: 26980801 Free PMC article.
References
-
- MacGlashan J, D. W, Bochner BS, Adelman DC, Jardieu PM, Togias A, Mckenzie-White J, et al. Down-regulation of FceRI expression on human basophils during in vivo treatment of atopic patients with anti-IgE antibody. J. Immunol. 1997;158:1438–45. - PubMed
-
- Prussin C, Griffith DT, Boesel KM, Lin H, Foster B, Casale TB. Omalizumab treatment downregulates dendritic cell FcepsilonRI expression. J Allergy Clin Immunol. 2003;112:1147–54. - PubMed
-
- MacGlashan D, Jr., Xia HZ, Schwartz LB, Gong J. IgE-regulated loss, not IgE-regulated synthesis, controls expression of FcepsilonRI in human basophils. J Leukoc Biol. 2001;70:207–18. - PubMed
-
- MacGlashan DW., Jr. Releasability of human basophils: Cellular sensitivity and maximal histamine release are independent variables. J Allergy Clin Immunol. 1993;91:605–15. - PubMed
-
- MacGlashan DW., Jr. Relationship Between Syk and SHIP Expression and Secretion from Human Basophils in the General Population. J. Allergy Clin. Immunol. 2007;119:626–33. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

