Adoptive transfer of tumor-specific Tc17 effector T cells controls the growth of B16 melanoma in mice
- PMID: 20237297
- PMCID: PMC2851479
- DOI: 10.4049/jimmunol.0902995
Adoptive transfer of tumor-specific Tc17 effector T cells controls the growth of B16 melanoma in mice
Abstract
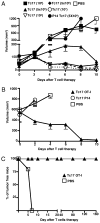
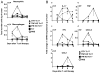
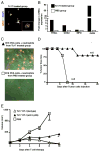
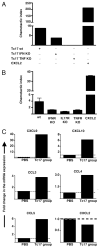
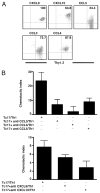
In vitro generated OVA-specific IL-17-producing CD8 T effector cells (Tc17) from OT-1 mice, adoptively transferred into B16-OVA tumor-bearing mice, controlled tumor growth in early and late stage melanoma. IL-17, TNF, and IFN-gamma from the Tc17 effectors all played a role in an enhanced recruitment of T cells, neutrophils, and macrophages to the tumor. In addition, Tc17 cells and recently recruited, activated neutrophils produced further chemokines, including CCL3, CCL4, CCL5, CXCL9, and CXCL10, responsible for the attraction of type 1 lymphocytes (Th1 and Tc1) and additional neutrophils. Neutrophils were rapidly attracted to the tumor site by an IL-17 dependent mechanism, but at later stages the induction of the chemokine CXCL2 by Tc17-derived TNF and IFN-gamma contributed to sustain neutrophil recruitment. Approximately 10-50 times as many Tc17 effectors were required compared with Tc1 effectors to exert the same level of control over tumor growth. The recruitment of neutrophils was more prominent when Tc17 rather than Tc1 were used to control tumor and depletion of neutrophils resulted in a diminished capacity to control tumor growth.
Conflict of interest statement
The authors have no conflicting financial interests.
Figures


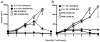
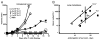
Similar articles
-
Adoptive transfer of Tc1 or Tc17 cells elicits antitumor immunity against established melanoma through distinct mechanisms.J Immunol. 2013 Feb 15;190(4):1873-81. doi: 10.4049/jimmunol.1201989. Epub 2013 Jan 11. J Immunol. 2013. PMID: 23315072 Free PMC article.
-
Tc1 and Tc2 effector cell therapy elicit long-term tumor immunity by contrasting mechanisms that result in complementary endogenous type 1 antitumor responses.J Immunol. 2004 Feb 1;172(3):1380-90. doi: 10.4049/jimmunol.172.3.1380. J Immunol. 2004. PMID: 14734713
-
Role of effector cell-derived IL-4, IL-5, and perforin in early and late stages of type 2 CD8 effector cell-mediated tumor rejection.J Immunol. 2001 Jul 1;167(1):424-34. doi: 10.4049/jimmunol.167.1.424. J Immunol. 2001. PMID: 11418679
-
Type 1 and type 2 CD8+ effector T cell subpopulations promote long-term tumor immunity and protection to progressively growing tumor.J Immunol. 2000 Jan 15;164(2):916-25. doi: 10.4049/jimmunol.164.2.916. J Immunol. 2000. PMID: 10623840
-
Multiple redundant effector mechanisms of CD8+ T cells protect against influenza infection.J Immunol. 2013 Jan 1;190(1):296-306. doi: 10.4049/jimmunol.1200571. Epub 2012 Nov 28. J Immunol. 2013. PMID: 23197262 Free PMC article.
Cited by
-
Integration of Salmonella into Combination Cancer Therapy.Cancers (Basel). 2021 Jun 28;13(13):3228. doi: 10.3390/cancers13133228. Cancers (Basel). 2021. PMID: 34203478 Free PMC article. Review.
-
PD-1 limits differentiation and plasticity of Tc17 cells.Front Immunol. 2023 Apr 28;14:1104730. doi: 10.3389/fimmu.2023.1104730. eCollection 2023. Front Immunol. 2023. PMID: 37205114 Free PMC article.
-
Friend or Foe - Tc17 cell generation and current evidence for their importance in human disease.Discov Immunol. 2023 Jul 20;2(1):kyad010. doi: 10.1093/discim/kyad010. eCollection 2023. Discov Immunol. 2023. PMID: 38567057 Free PMC article. Review.
-
Acute remote ischemic conditioning enhances (CD3+)- but not (FoxP3+)-T-cell invasion in the tumor center and increases IL 17 and TNF-alpha expression in a murine melanoma model.Front Immunol. 2024 Nov 22;15:1501885. doi: 10.3389/fimmu.2024.1501885. eCollection 2024. Front Immunol. 2024. PMID: 39650654 Free PMC article.
-
Linking tumor-associated macrophages, inflammation, and intestinal tumorigenesis: role of MCP-1.Am J Physiol Gastrointest Liver Physiol. 2012 Nov 15;303(10):G1087-95. doi: 10.1152/ajpgi.00252.2012. Epub 2012 Sep 27. Am J Physiol Gastrointest Liver Physiol. 2012. PMID: 23019193 Free PMC article.
References
-
- Chang AE, Aruga A, Cameron MJ, Sondak VK, Normolle DP, Fox BA, Shu S. Adoptive immunotherapy with vaccine-primed lymph node cells secondarily activated with anti-CD3 and interleukin-2. J Clin Oncol. 1997;15:796–807. - PubMed
-
- Chang AE, Yoshizawa H, Sakai K, Cameron MJ, Sondak VK, Shu S. Clinical observations on adoptive immunotherapy with vaccine-primed T-lymphocytes secondarily sensitized to tumor in vitro. Cancer Res. 1993;53:1043–1050. - PubMed
-
- Yee C, Thompson JA, Byrd D, Riddell SR, Roche P, Celis E, Greenberg PD. Adoptive T cell therapy using antigen-specific CD8+ T cell clones for the treatment of patients with metastatic melanoma: in vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci USA. 2002;99:16168–16173. - PMC - PubMed
-
- Mulé JJ, Shu S, Schwarz SL, Rosenberg SA. Adoptive immunotherapy of established pulmonary metastases with LAK cells and recombinant interleukin-2. Science. 1984;225:1487–1489. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

