Multiprotein complexes of Retinitis Pigmentosa GTPase regulator (RPGR), a ciliary protein mutated in X-linked Retinitis Pigmentosa (XLRP)
- PMID: 20238008
- PMCID: PMC3464500
- DOI: 10.1007/978-1-4419-1399-9_13
Multiprotein complexes of Retinitis Pigmentosa GTPase regulator (RPGR), a ciliary protein mutated in X-linked Retinitis Pigmentosa (XLRP)
Abstract
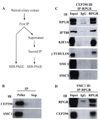
Mutations in Retinitis Pigmentosa GTPase Regulator (RPGR) are a frequent cause of X-linked Retinitis Pigmentosa (XLRP). The RPGR gene undergoes extensive alternative splicing and encodes for distinct protein isoforms in the retina. Extensive studies using isoform-specific antibodies and mouse mutants have revealed that RPGR predominantly localizes to the transition zone to primary cilia and associates with selected ciliary and microtubule-associated assemblies in photoreceptors. In this chapter, we have summarized recent advances on understanding the role of RPGR in photoreceptor protein trafficking. We also provide new evidence that suggests the existence of discrete RPGR multiprotein complexes in photoreceptors. Piecing together the RPGR-interactome in different subcellular compartments should provide critical insights into the role of alternative RPGR isoforms in associated orphan and syndromic retinal degenerative diseases.
Figures
Similar articles
-
Interaction of retinitis pigmentosa GTPase regulator (RPGR) with RAB8A GTPase: implications for cilia dysfunction and photoreceptor degeneration.Hum Mol Genet. 2010 Sep 15;19(18):3591-8. doi: 10.1093/hmg/ddq275. Epub 2010 Jul 14. Hum Mol Genet. 2010. PMID: 20631154 Free PMC article.
-
Retinitis Pigmentosa GTPase Regulator (RPGR) protein isoforms in mammalian retina: insights into X-linked Retinitis Pigmentosa and associated ciliopathies.Vision Res. 2008 Feb;48(3):366-76. doi: 10.1016/j.visres.2007.08.005. Epub 2007 Sep 27. Vision Res. 2008. PMID: 17904189 Free PMC article.
-
The Major Ciliary Isoforms of RPGR Build Different Interaction Complexes with INPP5E and RPGRIP1L.Int J Mol Sci. 2021 Mar 30;22(7):3583. doi: 10.3390/ijms22073583. Int J Mol Sci. 2021. PMID: 33808286 Free PMC article.
-
More Than Meets the Eye: Current Understanding of RPGR Function.Adv Exp Med Biol. 2018;1074:521-538. doi: 10.1007/978-3-319-75402-4_64. Adv Exp Med Biol. 2018. PMID: 29721984 Review.
-
Prevalence of RPGR-mutated X-linked retinitis pigmentosa among males.Ophthalmic Genet. 2022 Oct;43(5):581-588. doi: 10.1080/13816810.2022.2109686. Epub 2022 Aug 25. Ophthalmic Genet. 2022. PMID: 36004681 Review.
Cited by
-
Thicknesses of the retina and choroid in children with retinitis pigmentosa.BMC Ophthalmol. 2023 Jan 17;23(1):25. doi: 10.1186/s12886-023-02772-0. BMC Ophthalmol. 2023. PMID: 36650468 Free PMC article.
-
Tackling Primary Cilia Dysfunction in Photoreceptor Degenerative Diseases of the Eye.Int J Ophthalmic Pathol. 2012 Jun 24;1(1):e101. doi: 10.4172/2324-8599.1000e101. Int J Ophthalmic Pathol. 2012. PMID: 24455751 Free PMC article. No abstract available.
-
Systematic Identification of Candidate Genes for Inherited Retinal Disease Gene Therapy Integrating Worldwide IRD Cohort and Single-Cell Analysis.J Ophthalmol. 2025 Jun 12;2025:7014745. doi: 10.1155/joph/7014745. eCollection 2025. J Ophthalmol. 2025. PMID: 40547876 Free PMC article. Review.
-
Ablation of retinal ciliopathy protein RPGR results in altered photoreceptor ciliary composition.Sci Rep. 2015 Jun 11;5:11137. doi: 10.1038/srep11137. Sci Rep. 2015. PMID: 26068394 Free PMC article.
-
Ocular Ciliopathies: Genetic and Mechanistic Insights into Developing Therapies.Curr Med Chem. 2019;26(17):3120-3131. doi: 10.2174/0929867325666180917102557. Curr Med Chem. 2019. PMID: 30221600 Free PMC article. Review.
References
-
- Ayyagari R, Demirci FY, Liu J, et al. X-linked recessive atrophic macular degeneration from RPGR mutation. Genomics. 2002;80(2):166–171. - PubMed
-
- Badano JL, Mitsuma N, Beales PL, et al. The ciliopathies: an emerging class of human genetic disorders. Annu Rev Genomics Hum Genet. 2006;7:125–148. - PubMed
-
- Bartolini F, Bhamidipati A, Thomas S, et al. Functional overlap between retinitis pigmentosa 2 protein and the tubulin-specific chaperone cofactor C. J Biol Chem. 2002;277(17):14629–14634. - PubMed
-
- Beltran WA, Hammond P, Acland GM, et al. A frameshift mutation in RPGR exon ORF15 causes photoreceptor degeneration and inner retina remodeling in a model of X-linked retinitis pigmentosa. Invest Ophthalmol Vis Sci. 2006;47(4):1669–1681. - PubMed
-
- Besharse JC. The Retina: a model for cell biological studies Part I. New York: Academic; 1986. pp. 297–352.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

