Anti-infective activity of apolipoprotein domain derived peptides in vitro: identification of novel antimicrobial peptides related to apolipoprotein B with anti-HIV activity
- PMID: 20298574
- PMCID: PMC2855520
- DOI: 10.1186/1471-2172-11-13
Anti-infective activity of apolipoprotein domain derived peptides in vitro: identification of novel antimicrobial peptides related to apolipoprotein B with anti-HIV activity
Abstract
Background: Previous reports have shown that peptides derived from the apolipoprotein E receptor binding region and the amphipathic alpha-helical domains of apolipoprotein AI have broad anti-infective activity and antiviral activity respectively. Lipoproteins and viruses share a similar cell biological niche, being of overlapping size and displaying similar interactions with mammalian cells and receptors, which may have led to other antiviral sequences arising within apolipoproteins, in addition to those previously reported. We therefore designed a series of peptides based around either apolipoprotein receptor binding regions, or amphipathic alpha-helical domains, and tested these for antiviral and antibacterial activity.
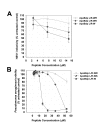
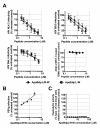
Results: Of the nineteen new peptides tested, seven showed some anti-infective activity, with two of these being derived from two apolipoproteins not previously used to derive anti-infective sequences. Apolipoprotein J (151-170) - based on a predicted amphipathic alpha-helical domain from apolipoprotein J - had measurable anti-HSV1 activity, as did apolipoprotein B (3359-3367) dp (apoBdp), the latter being derived from the LDL receptor binding domain B of apolipoprotein B. The more active peptide - apoBdp - showed similarity to the previously reported apoE derived anti-infective peptide, and further modification of the apoBdp sequence to align the charge distribution more closely to that of apoEdp or to introduce aromatic residues resulted in increased breadth and potency of activity. The most active peptide of this type showed similar potent anti-HIV activity, comparable to that we previously reported for the apoE derived peptide apoEdpL-W.
Conclusions: These data suggest that further antimicrobial peptides may be obtained using human apolipoprotein sequences, selecting regions with either amphipathic alpha-helical structure, or those linked to receptor-binding regions. The finding that an amphipathic alpha-helical region of apolipoprotein J has antiviral activity comparable with that for the previously reported apolipoprotein AI derived peptide 18A, suggests that full-length apolipoprotein J may also have such activity, as has been reported for full-length apolipoprotein AI. Although the strength of the anti-infective activity of the sequences identified was limited, this could be increased substantially by developing related mutant peptides. Indeed the apolipoprotein B-derived peptide mutants uncovered by the present study may have utility as HIV therapeutics or microbicides.
Figures



Similar articles
-
Apolipoprotein E-derived antimicrobial peptide analogues with altered membrane affinity and increased potency and breadth of activity.FEBS J. 2007 Sep;274(17):4511-25. doi: 10.1111/j.1742-4658.2007.05981.x. Epub 2007 Aug 6. FEBS J. 2007. PMID: 17681018
-
The receptor binding domain of apolipoprotein E, linked to a model class A amphipathic helix, enhances internalization and degradation of LDL by fibroblasts.Biochemistry. 2000 Jan 11;39(1):213-20. doi: 10.1021/bi991209w. Biochemistry. 2000. PMID: 10625496
-
Antiviral effects of apolipoprotein A-I and its synthetic amphipathic peptide analogs.Virology. 1990 May;176(1):48-57. doi: 10.1016/0042-6822(90)90229-k. Virology. 1990. PMID: 2158697
-
Apolipoprotein E mimetics and cholesterol-lowering properties.Am J Cardiovasc Drugs. 2011 Dec 1;11(6):371-81. doi: 10.2165/11594190-000000000-00000. Am J Cardiovasc Drugs. 2011. PMID: 22149316 Review.
-
Apolipoprotein/lipid interactions: studies with synthetic polypeptides.CRC Crit Rev Biochem. 1982;13(1):87-107. doi: 10.3109/10409238209108710. CRC Crit Rev Biochem. 1982. PMID: 6813024 Review.
Cited by
-
Human Cathelicidin Compensates for the Role of Apolipoproteins in Hepatitis C Virus Infectious Particle Formation.J Virol. 2016 Sep 12;90(19):8464-77. doi: 10.1128/JVI.00471-16. Print 2016 Oct 1. J Virol. 2016. PMID: 27440892 Free PMC article.
-
Peptide discovery across the spectrum of neuroinflammation; microglia and astrocyte phenotypical targeting, mediation, and mechanistic understanding.Front Mol Neurosci. 2024 Nov 20;17:1443985. doi: 10.3389/fnmol.2024.1443985. eCollection 2024. Front Mol Neurosci. 2024. PMID: 39634607 Free PMC article. Review.
-
Canine Bone Marrow Mesenchymal Stem Cell Conditioned Media Affect Bacterial Growth, Biofilm-Associated Staphylococcus aureus and AHL-Dependent Quorum Sensing.Microorganisms. 2020 Sep 26;8(10):1478. doi: 10.3390/microorganisms8101478. Microorganisms. 2020. PMID: 32993120 Free PMC article.
-
Proliferation and resistance difference of a liver-parasitized myxosporean in two different gynogenetic clones of gibel carp.Parasitol Res. 2014 Apr;113(4):1331-41. doi: 10.1007/s00436-014-3772-5. Epub 2014 Feb 1. Parasitol Res. 2014. PMID: 24488077
-
Impact of a Single Point Mutation on the Antimicrobial and Fibrillogenic Properties of Cryptides from Human Apolipoprotein B.Pharmaceuticals (Basel). 2021 Jun 29;14(7):631. doi: 10.3390/ph14070631. Pharmaceuticals (Basel). 2021. PMID: 34209895 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

