Identification and characterization of the two isoforms of the vertebrate H2A.Z histone variant
- PMID: 20299344
- PMCID: PMC2910051
- DOI: 10.1093/nar/gkq171
Identification and characterization of the two isoforms of the vertebrate H2A.Z histone variant
Abstract
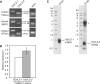
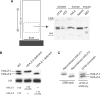
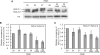
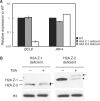
Histone variants play important roles in the epigenetic regulation of genome function. The histone variant H2A.Z is evolutionarily conserved from yeast to vertebrates, and it has been reported to have multiple effects upon gene expression and insulation, and chromosome segregation. Recently two genes encoding H2A.Z were identified in the vertebrate genome. However, it is not yet clear whether the proteins transcribed from these genes are functionally distinct. To address this issue, we knocked out each gene individually in chicken DT40 cells. We found that two distinct proteins, H2A.Z-1 and H2A.Z-2, were produced from these genes, and that these proteins could be separated on a long SDS-PAGE gel. The two isoforms were deposited to a similar extent by the SRCAP chromatin-remodeling complex, suggesting redundancy to their function. However, cells lacking either one of the two isoforms exhibited distinct alterations in cell growth and gene expression, suggesting that the two isoforms have differential effects upon nucleosome stability and chromatin structure. These findings provide insight into the molecular basis of the multiple functions of the H2A.Z gene products.
Figures


Similar articles
-
Patterning chromatin: form and function for H2A.Z variant nucleosomes.Curr Opin Genet Dev. 2006 Apr;16(2):119-24. doi: 10.1016/j.gde.2006.02.005. Epub 2006 Feb 28. Curr Opin Genet Dev. 2006. PMID: 16503125 Review.
-
Transcriptional and epigenetic functions of histone variant H2A.Z.Biochem Cell Biol. 2009 Feb;87(1):19-25. doi: 10.1139/O08-117. Biochem Cell Biol. 2009. PMID: 19234520 Review.
-
Reuniting the contrasting functions of H2A.Z.Biochem Cell Biol. 2006 Aug;84(4):528-35. doi: 10.1139/o06-077. Biochem Cell Biol. 2006. PMID: 16936825 Review.
-
Regulation of gene expression and cellular proliferation by histone H2A.Z.Biochem Cell Biol. 2009 Feb;87(1):179-88. doi: 10.1139/O08-138. Biochem Cell Biol. 2009. PMID: 19234533 Review.
-
Quickly evolving histones, nucleosome stability and chromatin folding: all about histone H2A.Bbd.Gene. 2008 Apr 30;413(1-2):1-7. doi: 10.1016/j.gene.2008.02.003. Epub 2008 Feb 16. Gene. 2008. PMID: 18329190 Review.
Cited by
-
Non-redundant functions of H2A.Z.1 and H2A.Z.2 in chromosome segregation and cell cycle progression.EMBO Rep. 2021 Nov 4;22(11):e52061. doi: 10.15252/embr.202052061. Epub 2021 Aug 23. EMBO Rep. 2021. PMID: 34423893 Free PMC article.
-
Deposition of histone variant H2A.Z within gene bodies regulates responsive genes.PLoS Genet. 2012;8(10):e1002988. doi: 10.1371/journal.pgen.1002988. Epub 2012 Oct 11. PLoS Genet. 2012. PMID: 23071449 Free PMC article.
-
SUMO modification system facilitates the exchange of histone variant H2A.Z-2 at DNA damage sites.Nucleus. 2018 Jan 1;9(1):87-94. doi: 10.1080/19491034.2017.1395543. Epub 2017 Dec 14. Nucleus. 2018. PMID: 29095668 Free PMC article.
-
Histone variants: emerging players in cancer biology.Cell Mol Life Sci. 2014 Feb;71(3):379-404. doi: 10.1007/s00018-013-1343-z. Epub 2013 May 8. Cell Mol Life Sci. 2014. PMID: 23652611 Free PMC article. Review.
-
Brain-specific deletion of histone variant H2A.z results in cortical neurogenesis defects and neurodevelopmental disorder.Nucleic Acids Res. 2018 Mar 16;46(5):2290-2307. doi: 10.1093/nar/gkx1295. Nucleic Acids Res. 2018. PMID: 29294103 Free PMC article.
References
-
- Luger K, Rechsteiner TJ, Flaus AJ, Waye MM, Richmond TJ. Characterization of nucleosome core particles containing histone proteins made in bacteria. J. Mol. Biol. 1997;272:301–311. - PubMed
-
- Kornberg RD, Lorch Y. Twenty-five years of the nucleosome, fundamental particle of the eukaryote chromosome. Cell. 1999;98:285–294. - PubMed
-
- Altaf M, Auger A, Covic M, Côté J. Connection between histone H2A variants and chromatin remodeling complexes. Biochem. Cell Biol. 2009;87:35–50. - PubMed
-
- Santenard A, Torres-Padilla ME. Epigenetic reprogramming in mammalian reproduction: contribution from histone variants. Epigenetics. 2009;4:80–84. - PubMed
-
- Ren Q, Gorovsky MA. Histone H2A.Z acetylation modulates an essential charge patch. Mol. Cell. 2001;7:1329–1335. - PubMed

