Identification of Rhodococcus equi lipids recognized by host cytotoxic T lymphocytes
- PMID: 20299405
- PMCID: PMC4086205
- DOI: 10.1099/mic.0.035915-0
Identification of Rhodococcus equi lipids recognized by host cytotoxic T lymphocytes
Abstract
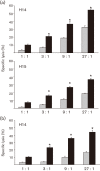
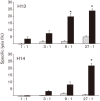
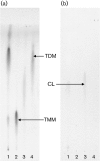
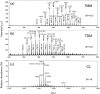
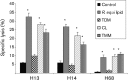
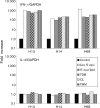
Immune adult horses have CD8(+) cytotoxic T lymphocytes (CTLs) that recognize and lyse Rhodococcus equi-infected cells in an equine lymphocyte alloantigen (ELA)-A [classical major histocompatibility complex (MHC) class I]-unrestricted fashion. As protein antigens are MHC class I-restricted, the lack of restriction suggests that the bacterial antigens being recognized by the host are not proteins. The goals of this study were to test the hypothesis that these CTLs recognize unique R. equi cell-wall lipids related to mycobacterial lipids. Initial experiments showed that treatment of soluble R. equi antigen with broadly reactive proteases did not significantly diminish the ability of the antigen to stimulate R. equi-specific CTLs. R. equi-specific CTLs were also shown to lyse target cells (equine macrophages) pulsed with an R. equi lipid extract. Analysis of the R. equi lipid by TLC and MS (MALDI-TOF and ES) indicated that the extracted antigen consisted of three primary fractions: trehalose monomycolate (TMM), trehalose dimycolate (TDM) and cardiolipin (CL). ELA-A-mismatched cells pulsed with purified TMM and CL, but not the TDM fraction, were recognized and lysed by R. equi-specific CTLs. Because of their role in immune clearance and pathogenesis, transcription of the cytokines gamma interferon (IFN-gamma) and interleukin-4 (IL-4) was also measured in response to R. equi lipids by using real-time PCR; elevated IFN-gamma, but not IL-4, was associated with host clearance of the bacteria. The whole-cell R. equi lipid and all three R. equi lipid fractions resulted in marked increases in IFN-gamma transcription, but no increase in IL-4 transcription. Together, these data support the hypothesis that immune recognition of unique lipids in the bacterial cell wall is an important component of the protective immune response to R. equi. The results also identify potential lipid antigens not previously shown to be recognized by CTLs in an important, naturally occurring actinomycete bacterial pathogen.
Figures

Similar articles
-
Rhodococcus equi-specific cytotoxic T lymphocytes in immune horses and development in asymptomatic foals.Infect Immun. 2005 Apr;73(4):2083-93. doi: 10.1128/IAI.73.4.2083-2093.2005. Infect Immun. 2005. PMID: 15784549 Free PMC article.
-
Rhodococcus equi-infected macrophages are recognized and killed by CD8+ T lymphocytes in a major histocompatibility complex class I-unrestricted fashion.Infect Immun. 2004 Dec;72(12):7073-83. doi: 10.1128/IAI.72.12.7073-7083.2004. Infect Immun. 2004. PMID: 15557631 Free PMC article.
-
Experimental infection of neonatal foals with Rhodococcus equi triggers adult-like gamma interferon induction.Clin Vaccine Immunol. 2007 Jun;14(6):669-77. doi: 10.1128/CVI.00042-07. Epub 2007 Apr 4. Clin Vaccine Immunol. 2007. PMID: 17409222 Free PMC article.
-
Cytotoxic T lymphocytes in protection against equine infectious anemia virus.Anim Health Res Rev. 2004 Dec;5(2):271-6. doi: 10.1079/ahr200482. Anim Health Res Rev. 2004. PMID: 15984338 Review.
-
Molecular and infection biology of the horse pathogen Rhodococcus equi.FEMS Microbiol Rev. 2009 Sep;33(5):870-91. doi: 10.1111/j.1574-6976.2009.00181.x. Epub 2009 Apr 23. FEMS Microbiol Rev. 2009. PMID: 19453748 Review.
Cited by
-
The equine CD1 gene family is the largest and most diverse yet identified.Immunogenetics. 2014 Jan;66(1):33-42. doi: 10.1007/s00251-013-0741-6. Epub 2013 Nov 7. Immunogenetics. 2014. PMID: 24196432
-
A conserved human CD4+ T cell subset recognizing the mycobacterial adjuvant trehalose monomycolate.J Clin Invest. 2024 Dec 24;135(6):e185443. doi: 10.1172/JCI185443. J Clin Invest. 2024. PMID: 39718834 Free PMC article.
-
Diversion of phagosome trafficking by pathogenic Rhodococcus equi depends on mycolic acid chain length.Cell Microbiol. 2013 Mar;15(3):458-73. doi: 10.1111/cmi.12050. Epub 2012 Nov 13. Cell Microbiol. 2013. PMID: 23078612 Free PMC article.
-
Early development of cytotoxic T lymphocytes in neonatal foals following oral inoculation with Rhodococcus equi.Vet Immunol Immunopathol. 2011 Jun 15;141(3-4):312-6. doi: 10.1016/j.vetimm.2011.03.015. Epub 2011 Mar 21. Vet Immunol Immunopathol. 2011. PMID: 21481947 Free PMC article.
-
Effects of administration of live or inactivated virulent Rhodococccus equi and age on the fecal microbiome of neonatal foals.PLoS One. 2013 Jun 13;8(6):e66640. doi: 10.1371/journal.pone.0066640. Print 2013. PLoS One. 2013. PMID: 23785508 Free PMC article.
References
-
- Beckman E. M., Porcelli S. A., Morita C. T., Behar S. M., Furlong S. T., Brenner M. B. (1994). Recognition of a lipid antigen by CD1-restricted αβ+ T cells. Nature 372, 691–694. - PubMed
-
- Bhatt A., Fujiwara N., Bhatt K., Gurcha S. S., Kremer L., Chen B., Chan J., Porcelli S. A., Kobayashi K. & other authors (2007). Deletion of kasB in Mycobacterium tuberculosis causes loss of acid-fastness and subclinical latent tuberculosis in immunocompetent mice. Proc Natl Acad Sci U S A 104, 5157–5162. - PMC - PubMed
-
- Boom W. H., Canaday D. H., Fulton S. A., Gehring A. J., Rojas R. E., Torres M. (2003). Human immunity to M. tuberculosis: T cell subsets and antigen processing. Tuberculosis (Edinb) 83, 98–106. - PubMed
-
- Brennan P. J., Nikaido H. (1995). The envelope of mycobacteria. Annu Rev Biochem 64, 29–63. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

