Dicalcin inhibits fertilization through its binding to a glycoprotein in the egg envelope in Xenopus laevis
- PMID: 20299459
- PMCID: PMC2865274
- DOI: 10.1074/jbc.M109.079483
Dicalcin inhibits fertilization through its binding to a glycoprotein in the egg envelope in Xenopus laevis
Abstract
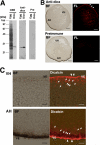
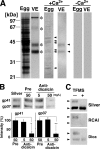
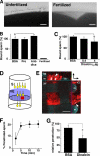
Fertilization comprises oligosaccharide-mediated sperm-egg interactions, including sperm binding to an extracellular egg envelope, sperm penetration through the envelope, and fusion with an egg plasma membrane. We show that Xenopus dicalcin, an S100-like Ca(2+)-binding protein, present in the extracellular egg envelope (vitelline envelope (VE)), is a suppressive mediator of sperm-egg interaction. Preincubation with specific antibody greatly increased the efficiency of in vitro fertilization, whereas prior application of exogenous dicalcin substantially inhibited fertilization as well as sperm binding to an egg and in vitro sperm penetration through the VE protein layer. Dicalcin showed binding to protein cores of gp41 and gp37, constituents of VE, in a Ca(2+)-dependent manner and increased in vivo reactivity of VE with a lectin, Ricinus communis agglutinin I, which was accounted for by increased binding ability of gp41 to the lectin and greater exposure of gp41 to an external environment. Our findings strongly suggest that dicalcin regulates the distribution of oligosaccharides within the VE through its binding to the protein core of gp41, probably by modulating configuration of oligosaccharides on gp41 and the three-dimensional structure of VE framework, and thereby plays a pivotal role in sperm-egg interactions during fertilization.
Figures
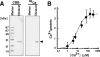


References
-
- Yanagimachi R. (1994) in The Physiology of Reproduction (Knobil E., Neill J. D. eds) pp. 189–317, Raven Press, New York
-
- Primakoff P., Myles D. G. (2002) Science 296, 2183–2185 - PubMed
-
- Hoodbhoy T., Dean J. (2004) Reproduction 127, 417–422 - PubMed
-
- Litscher E. S., Wassarman P. M. (2007) Histol. Histopathol. 22, 337–347 - PubMed
-
- Clark G. F., Dell A. (2006) J. Biol. Chem. 281, 13853–13856 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

