DNIC-mediated analgesia produced by a supramaximal electrical or a high-dose formalin conditioning stimulus: roles of opioid and alpha2-adrenergic receptors
- PMID: 20302612
- PMCID: PMC2850336
- DOI: 10.1186/1423-0127-17-19
DNIC-mediated analgesia produced by a supramaximal electrical or a high-dose formalin conditioning stimulus: roles of opioid and alpha2-adrenergic receptors
Abstract
Background: Diffuse noxious inhibitory controls (DNIC) can be produced by different types of conditioning stimuli, but the analgesic properties and underlying mechanisms remain unclear. The aim of this study was to differentiate the induction of DNIC analgesia between noxious electrical and inflammatory conditioning stimuli.
Methods: First, rats subjected to either a supramaximal electrical stimulation or an injection of high-dose formalin in the hind limb were identified to have pain responses with behavioral evidence and spinal Fos-immunoreactive profiles. Second, suppression of tail-flick latencies by the two noxious stimuli was assessed to confirm the presence of DNIC. Third, an opioid receptor antagonist (naloxone) and an alpha2-adrenoreceptor antagonist (yohimbine) were injected, intraperitoneally and intrathecally respectively, before conditioning noxious stimuli to test the involvement of descending inhibitory pathways in DNIC-mediated analgesia.
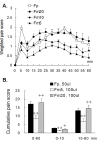
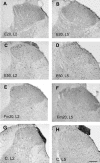
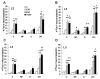
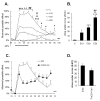
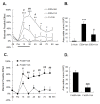
Results: An intramuscular injection of 100 microl of 5% formalin produced noxious behaviors with cumulative pain scores similar to those of 50 microl of 2% formalin in the paw. Both electrical and chemical stimulation significantly increased Fos expression in the superficial dorsal horns, but possessed characteristic distribution patterns individually. Both conditioning stimuli prolonged the tail-flick latencies indicating a DNIC response. However, the electrical stimulation-induced DNIC was reversed by yohimbine, but not by naloxone; whereas noxious formalin-induced analgesia was both naloxone- and yohimbine-reversible.
Conclusions: It is demonstrated that DNIC produced by different types of conditioning stimuli can be mediated by different descending inhibitory controls, indicating the organization within the central nervous circuit is complex and possibly exhibits particular clinical manifestations.
Figures

Similar articles
-
Blockade of opioid receptors in the medullary reticularis nucleus dorsalis, but not the rostral ventromedial medulla, prevents analgesia produced by diffuse noxious inhibitory control in rats with muscle inflammation.J Pain. 2011 Jun;12(6):687-97. doi: 10.1016/j.jpain.2010.12.009. Epub 2011 Feb 17. J Pain. 2011. PMID: 21330219 Free PMC article.
-
Impaired diffuse noxious inhibitory controls in specific alternation of rhythm in temperature-stressed rats.Eur J Pharmacol. 2016 Aug 5;784:61-8. doi: 10.1016/j.ejphar.2016.05.011. Epub 2016 May 10. Eur J Pharmacol. 2016. PMID: 27178898
-
NK1 receptor-expressing spinoparabrachial neurons trigger diffuse noxious inhibitory controls through lateral parabrachial activation in the male rat.Pain. 2009 Apr;142(3):245-254. doi: 10.1016/j.pain.2009.01.015. Epub 2009 Feb 23. Pain. 2009. PMID: 19231081
-
The activation of bulbo-spinal controls by peripheral nociceptive inputs: diffuse noxious inhibitory controls.Biol Res. 1995;28(1):113-25. Biol Res. 1995. PMID: 8728826 Review.
-
Diffuse noxious inhibitory controls (DNIC) in animals and in man.Patol Fiziol Eksp Ter. 1992 Jul-Aug;(4):55-65. Patol Fiziol Eksp Ter. 1992. PMID: 1303506 Review.
Cited by
-
Reduction of conditioned pain modulation in humans by naltrexone: an exploratory study of the effects of pain catastrophizing.J Behav Med. 2013 Jun;36(3):315-27. doi: 10.1007/s10865-012-9424-2. Epub 2012 Apr 26. J Behav Med. 2013. PMID: 22534819 Free PMC article. Clinical Trial.
-
Individual differences in acute pain-induced endogenous analgesia predict time to resolution of postoperative pain in the rat.Anesthesiology. 2015 Apr;122(4):895-907. doi: 10.1097/ALN.0000000000000593. Anesthesiology. 2015. PMID: 25581910 Free PMC article.
-
Involvement of spinal α2 -adrenoceptors in prolonged modulation of hind limb withdrawal reflexes following acute noxious stimulation in the anaesthetized rabbit.Eur J Neurosci. 2016 Mar;43(6):834-45. doi: 10.1111/ejn.13185. Epub 2016 Feb 28. Eur J Neurosci. 2016. PMID: 26804327 Free PMC article.
-
Pain inhibition-the unintended benefit of electrically elicited muscle strengthening contractions.BMC Musculoskelet Disord. 2023 Feb 18;24(1):131. doi: 10.1186/s12891-023-06243-x. BMC Musculoskelet Disord. 2023. PMID: 36803339 Free PMC article.
-
A study of cortical and brainstem mechanisms of diffuse noxious inhibitory controls in anaesthetised normal and neuropathic rats.Eur J Neurosci. 2020 Feb;51(4):952-962. doi: 10.1111/ejn.14576. Epub 2019 Oct 6. Eur J Neurosci. 2020. PMID: 31518451 Free PMC article.
References
-
- Hu JW. Response properties of nociceptive and non-nociceptive neurons in the rat's trigeminal subnucleus caudalis (medullary dorsal horn) related to cutaneous and deep craniofacial afferent stimulation and modulation by diffuse noxious inhibitory controls. Pain. 1990;41:331–345. doi: 10.1016/0304-3959(90)90010-B. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

