Repression of beta-catenin signaling by PPAR gamma ligands
- PMID: 20303941
- PMCID: PMC2866743
- DOI: 10.1016/j.ejphar.2010.03.010
Repression of beta-catenin signaling by PPAR gamma ligands
Abstract
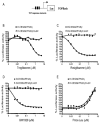
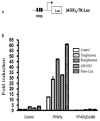
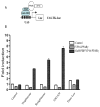
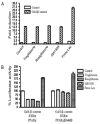
Aberrant activation of the Wnt/beta-catenin signaling pathway plays a crucial role in oncogenesis of various human malignancies. It has been demonstrated that there is a direct interaction between beta-catenin and PPAR gamma. Here we examined the effects of fifteen reported PPAR ligands in a reporter gene assay that is dependent on beta-catenin activation of TCF/LEF transcription factors; only the thiazolidinedione PPAR gamma agonists troglitazone, rosiglitazone and pioglitazone, and a non-thiazolidinedione PPAR gamma activator GW1929 inhibited beta-catenin-induced transcription in a PPAR gamma dependent fashion. The results from mammalian one-hybrid experiments showed that functional PPAR gamma was necessary for ligand-dependent inhibition of beta-catenin transactivation. However, a PPAR gamma activator Fmoc-Leu could not repress beta-catenin-mediated signaling and its transactivation activity. These results indicate that activation of PPAR gamma is necessary, but not sufficient, for the beta-catenin antagonistic activity of a PPAR gamma agonist, and that the inhibitory compounds interfere directly with beta-catenin transactivation activity.
(c) 2010 Elsevier B.V. All rights reserved.
Figures
References
-
- Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006;127:469–480. - PubMed
-
- Crawford HC, Fingleton BM, Rudolph-Owen LA, Goss KJ, Rubinfeld B, Polakis P, Matrisian LM. The metalloproteinase matrilysin is a target of beta-catenin transactivation in intestinal tumors. Oncogene. 1999;18:2883–2891. - PubMed
-
- Debril MB, Renaud JP, Fajas L, Auwerx J. The pleiotropic functions of peroxisome proliferator-activated receptor gamma. Journal of molecular medicine (Berlin, Germany) 2001;79:30–47. - PubMed
-
- Forman BM, Tontonoz P, Chen J, Brun RP, Spiegelman BM, Evans RM. 15-Deoxy-delta 12, 14-prostaglandin J2 is a ligand for the adipocyte determination factor PPAR gamma. Cell. 1995;83:803–812. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

