Compounds that target host cell proteins prevent varicella-zoster virus replication in culture, ex vivo, and in SCID-Hu mice
- PMID: 20307580
- PMCID: PMC2866756
- DOI: 10.1016/j.antiviral.2010.03.007
Compounds that target host cell proteins prevent varicella-zoster virus replication in culture, ex vivo, and in SCID-Hu mice
Abstract
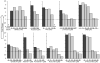
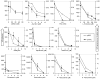
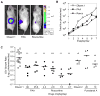
Varicella-zoster virus (VZV) replicates in quiescent T cells, neurons, and skin cells. In cultured fibroblasts (HFFs), VZV induces host cyclin expression and cyclin-dependent kinase (CDK) activity without causing cell cycle progression. CDK1/cyclin B1 phosphorylates the major viral transactivator, and the CDK inhibitor roscovitine prevents VZV mRNA transcription. We investigated the antiviral effects of additional compounds that target CDKs or other cell cycle enzymes in culture, ex vivo, and in vivo. Cytotoxicity and cell growth arrest doses were determined by Neutral Red assay. Antiviral effects were evaluated in HFFs by plaque assay, genome copy number, and bioluminescence. Positive controls were acyclovir (400 microM) and phosphonoacetic acid (PAA, 1 mM). Test compounds were roscovitine, aloisine A, and purvalanol A (CDK inhibitors), aphidicolin (inhibits human and herpesvirus DNA polymerase), l-mimosine (indirectly inhibits human DNA polymerase), and DRB (inhibits casein kinase 2). All had antiviral effects below the concentrations required for cell growth arrest. Compounds were tested in skin organ culture at EC(99) doses; all prevented VZV replication in skin, except for aloisine A and purvalanol A. In SCID mice with skin xenografts, roscovitine (0.7 mg/kg/day) was as effective as PAA (36 mg/kg/day). The screening systems described here are useful models for evaluating novel antiviral drugs for VZV.
Copyright 2010 Elsevier B.V. All rights reserved.
Figures

Similar articles
-
A Novel Human Skin Tissue Model To Study Varicella-Zoster Virus and Human Cytomegalovirus.J Virol. 2020 Oct 27;94(22):e01082-20. doi: 10.1128/JVI.01082-20. Print 2020 Oct 27. J Virol. 2020. PMID: 32878893 Free PMC article.
-
Roscovitine, a cyclin-dependent kinase inhibitor, prevents replication of varicella-zoster virus.J Virol. 2004 Mar;78(6):2853-62. doi: 10.1128/jvi.78.6.2853-2862.2004. J Virol. 2004. PMID: 14990704 Free PMC article.
-
β-l-1-[5-(E-2-bromovinyl)-2-(hydroxymethyl)-1,3-(dioxolan-4-yl)] uracil (l-BHDU) prevents varicella-zoster virus replication in a SCID-Hu mouse model and does not interfere with 5-fluorouracil catabolism.Antiviral Res. 2014 Oct;110:10-9. doi: 10.1016/j.antiviral.2014.07.007. Epub 2014 Jul 19. Antiviral Res. 2014. PMID: 25051026 Free PMC article.
-
Viral and cellular kinases are potential antiviral targets and have a central role in varicella zoster virus pathogenesis.Biochim Biophys Acta. 2004 Mar 11;1697(1-2):225-31. doi: 10.1016/j.bbapap.2003.11.026. Biochim Biophys Acta. 2004. PMID: 15023363 Review.
-
Effects of varicella-zoster virus on cell cycle regulatory pathways.Curr Top Microbiol Immunol. 2010;342:67-77. doi: 10.1007/82_2010_28. Curr Top Microbiol Immunol. 2010. PMID: 20397072 Review.
Cited by
-
Humanized Severe Combined Immunodeficient (SCID) Mouse Models for Varicella-Zoster Virus Pathogenesis.Curr Top Microbiol Immunol. 2023;438:135-161. doi: 10.1007/82_2022_255. Curr Top Microbiol Immunol. 2023. PMID: 35292858
-
CK2 inhibitors increase the sensitivity of HSV-1 to interferon-β.Antiviral Res. 2011 Sep;91(3):259-66. doi: 10.1016/j.antiviral.2011.06.009. Epub 2011 Jun 22. Antiviral Res. 2011. PMID: 21722672 Free PMC article.
-
Potential of protein kinase inhibitors for treating herpesvirus-associated disease.Trends Microbiol. 2013 Jun;21(6):286-95. doi: 10.1016/j.tim.2013.03.005. Epub 2013 Apr 19. Trends Microbiol. 2013. PMID: 23608036 Free PMC article. Review.
-
Dynamic changes in protein interaction between AKAP95 and Cx43 during cell cycle progression of A549 cells.Sci Rep. 2016 Feb 16;6:21224. doi: 10.1038/srep21224. Sci Rep. 2016. PMID: 26880274 Free PMC article.
-
A Novel Human Skin Tissue Model To Study Varicella-Zoster Virus and Human Cytomegalovirus.J Virol. 2020 Oct 27;94(22):e01082-20. doi: 10.1128/JVI.01082-20. Print 2020 Oct 27. J Virol. 2020. PMID: 32878893 Free PMC article.
References
-
- Babich H, Sedletcaia A, Kenigsberg B. In vitro cytotoxicity of protocatechuic acid to cultured human cells from oral tissue: involvement in oxidative stress. Pharmacol Toxicol. 2002;91:245–253. - PubMed
-
- Bresnahan WA, Boldogh I, Chi P, Thompson EA, Albrecht T. Inhibition of cellular Cdk2 activity blocks human cytomegalovirus replication. Virology. 1997;231:239–247. - PubMed
-
- Cohen JI, Sato H, Srinivas S, Lekstrom K. Varicella-zoster virus (VZV) ORF65 virion protein is dispensable for replication in cell culture and is phosphorylated by casein kinase II, but not by the VZV protein kinases. Virology. 2001;280:62–71. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

