Ion-dependent polymerization differences between mammalian beta- and gamma-nonmuscle actin isoforms
- PMID: 20308063
- PMCID: PMC2871477
- DOI: 10.1074/jbc.M110.110130
Ion-dependent polymerization differences between mammalian beta- and gamma-nonmuscle actin isoforms
Abstract
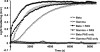
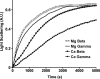
beta- and gamma-nonmuscle actins differ by 4 amino acids at or near the N terminus and distant from polymerization interfaces. beta-Actin contains an Asp(1)-Asp(2)-Asp(3) and Val(10) whereas gamma-actin has a Glu(1)-Glu(2)-Glu(3) and Ile(10). Despite these small changes, conserved across mammals, fish, and birds, their differential localization in the same cell suggests they may play different roles reflecting differences in their biochemical properties. To test this hypothesis, we established a baculovirus-driven expression system for producing these actins in isoform-pure populations although contaminated with 20-25% insect actin. Surprisingly, Ca-gamma-actin exhibits a slower monomeric nucleotide exchange rate, a much longer nucleation phase, and a somewhat slower elongation rate than beta-actin. In the Mg-form, this difference between the two is much smaller. Ca-gamma-actin depolymerizes half as fast as does beta-actin. Mixing experiments with Ca-actins reveal the two will readily co-polymerize. In the Ca-form, phosphate release from polymerizing beta-actin occurs much more rapidly and extensively than polymerization, whereas phosphate release lags behind polymerization with gamma-actin. Phosphate release during treadmilling is twice as fast with beta- as with gamma-actin. With Mg-actin in the initial stages, phosphate release for both actins correlates much more closely with polymerization. Calcium bound in the high affinity binding site of gamma-actin may cause a selective energy barrier relative to beta-actin that retards the equilibration between G- and F-monomer conformations resulting in a slower polymerizing actin with greater filament stability. This difference may be particularly important in sites such as the gamma-actin-rich cochlear hair cell stereocilium where local mm calcium concentrations may exist.
Figures

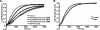


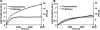
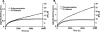
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

