Gonadotropin-positive pituitary tumors accompanied by ovarian tumors in aging female ERbeta-/- mice
- PMID: 20308571
- PMCID: PMC2851950
- DOI: 10.1073/pnas.1002029107
Gonadotropin-positive pituitary tumors accompanied by ovarian tumors in aging female ERbeta-/- mice
Abstract
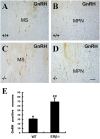
At 2 years of age, 100% (23/23) of ERbeta(-/-) female mice have developed large pituitary and ovarian tumors. The pituitary tumors are gonadotropin-positive and the ovarian tumors are sex cord (less differentiated) and granulosa cell tumors (differentiated and estrogen secreting). No male mice had pituitary tumors and no pituitary or ovarian tumors developed in ERalpha(-/-) mice or in ERalphabeta(-/-) double knockout mice. The tumors have high proliferation indices, are ERalpha-positive, ERbeta-negative, and express high levels of nuclear phospho-SMAD3. Mice with granulosa cell tumors also had hyperproliferative endometria. The cause of the pituitary tumors appeared to be excessive secretion of gonadotropin releasing hormone (GnRH) from the hypothalamus resulting from high expression of NPY. The ovarian phenotype is similar to that seen in mice where inhibin is ablated. The data indicate that ERbeta plays an important role in regulating GnRH secretion. We suggest that in the absence of ERbeta, the proliferative action of FSH/SMAD3 is unopposed and the high proliferation leads to the development of ovarian tumors. The absence of tumors in the ERalphabeta(-/-) mice suggests that tumor development requires the presence of ERalpha.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
The bi-modal effects of estradiol on gonadotropin synthesis and secretion in female mice are dependent on estrogen receptor-alpha.J Endocrinol. 2006 Oct;191(1):309-17. doi: 10.1677/joe.1.06965. J Endocrinol. 2006. PMID: 17065413
-
Sexually dimorphic roles of steroid hormone receptor signaling in gonadal tumorigenesis.Mol Endocrinol. 2003 Oct;17(10):2039-52. doi: 10.1210/me.2003-0039. Epub 2003 Jul 10. Mol Endocrinol. 2003. PMID: 12855748
-
Prevention of the polycystic ovarian phenotype and characterization of ovulatory capacity in the estrogen receptor-alpha knockout mouse.Endocrinology. 1999 Dec;140(12):5855-65. doi: 10.1210/endo.140.12.7222. Endocrinology. 1999. PMID: 10579351
-
Selective expression of estrogen receptor alpha and beta isoforms in human pituitary tumors.J Clin Endocrinol Metab. 1998 Nov;83(11):3965-72. doi: 10.1210/jcem.83.11.5236. J Clin Endocrinol Metab. 1998. PMID: 9814476 Clinical Trial.
-
PES1 differentially regulates the expression of ERα and ERβ in ovarian cancer.IUBMB Life. 2013 Dec;65(12):1017-25. doi: 10.1002/iub.1228. Epub 2013 Nov 24. IUBMB Life. 2013. PMID: 24376209
Cited by
-
Role of estrogen receptor α and β in preserving hippocampal function during aging.J Neurosci. 2013 Feb 6;33(6):2671-83. doi: 10.1523/JNEUROSCI.4937-12.2013. J Neurosci. 2013. PMID: 23392694 Free PMC article.
-
Insights into granulosa cell tumors using spontaneous or genetically engineered mouse models.Clin Exp Reprod Med. 2016 Mar;43(1):1-8. doi: 10.5653/cerm.2016.43.1.1. Epub 2016 Mar 31. Clin Exp Reprod Med. 2016. PMID: 27104151 Free PMC article. Review.
-
Characterizing Endocrine Status, Tumor Hypoxia and Immunogenicity for Therapy Success in Epithelial Ovarian Cancer.Front Endocrinol (Lausanne). 2021 Nov 17;12:772349. doi: 10.3389/fendo.2021.772349. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34867818 Free PMC article. Review.
-
ERβ in Granulosa Cell Tumors and Its Clinical Potential.Endocrinology. 2023 Apr 17;164(6):bqad063. doi: 10.1210/endocr/bqad063. Endocrinology. 2023. PMID: 37075218 Free PMC article. Review.
-
Minireview: animal models and mechanisms of ovarian cancer development.Endocrinology. 2012 Apr;153(4):1585-92. doi: 10.1210/en.2011-2121. Epub 2012 Mar 6. Endocrinology. 2012. PMID: 22396450 Free PMC article. Review.
References
-
- Gharib SD, Wierman ME, Shupnik MA, Chin WW. Molecular biology of the pituitary gonadotropins. Endocr Rev. 1990;11:177–199. - PubMed
-
- Petersen SL, Ottem EN, Carpenter CD. Direct and indirect regulation of gonadotropin-releasing hormone neurons by estradiol. Biol Reprod. 2003;69:1771–1778. - PubMed
-
- Shupnik MA, Gharib SD, Chin WW. Estrogen suppresses rat gonadotropin gene transcription in vivo. Endocrinology. 1988;122:1842–1846. - PubMed
-
- Burger LL, Haisenleder DJ, Dalkin AC, Marshall JC. Regulation of gonadotropin subunit gene transcription. J Mol Endocrinol. 2004;33:559–584. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

