Phase II trial of bevacizumab, thalidomide, docetaxel, and prednisone in patients with metastatic castration-resistant prostate cancer
- PMID: 20308663
- PMCID: PMC2860408
- DOI: 10.1200/JCO.2009.25.4524
Phase II trial of bevacizumab, thalidomide, docetaxel, and prednisone in patients with metastatic castration-resistant prostate cancer
Abstract
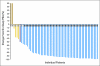
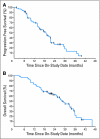
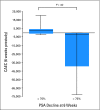
PURPOSE We previously demonstrated that thalidomide appears to add to the activity of docetaxel in metastatic castration-resistant prostate cancer (CRPC). Phase II studies combining docetaxel with bevacizumab have had substantial antitumor activity. We hypothesized that the combination of docetaxel plus these antiangiogenic drugs with different targets would have substantial clinical activity. To explore safety and efficacy, this was tested in mice and in human patients. PATIENTS AND METHODS Preclinical efficacy of the combination therapy was evaluated in PC3 xenograft mice. Sixty patients with progressive metastatic CRPC received intravenous docetaxel and bevacizumab plus oral thalidomide and prednisone. The primary end point was a prostate-specific antigen (PSA) decline of > or = 50%. Secondary end points included time to progression, overall survival, and safety. Results In the mouse model, combination therapy of docetaxel, bevacizumab, and thalidomide inhibited tumor growth most effectively. In the clinical trial, 90% of patients receiving the combination therapy had PSA declines of > or = 50%, and 88% achieved a PSA decline of > or = 30% within the first 3 months of treatment. The median time to progression was 18.3 months, and the median overall survival was 28.2 months in this group with a Halabi-predicted survival of 14 months. While toxicities were manageable, all patients developed grade 3/4 neutropenia. CONCLUSION The addition of bevacizumab and thalidomide to docetaxel is a highly active combination with manageable toxicities. The estimated median survival is encouraging, given the generally poor prognosis of this patient population. These results suggest that definitive clinical trials combining antiangiogenic agent combinations with docetaxel are warranted to improve treatment outcomes for patients with metastatic CRPC.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures

Similar articles
-
Randomized, double-blind, placebo-controlled phase III trial comparing docetaxel and prednisone with or without bevacizumab in men with metastatic castration-resistant prostate cancer: CALGB 90401.J Clin Oncol. 2012 May 1;30(13):1534-40. doi: 10.1200/JCO.2011.39.4767. Epub 2012 Mar 26. J Clin Oncol. 2012. PMID: 22454414 Free PMC article. Clinical Trial.
-
A phase 2 study of estramustine, docetaxel, and bevacizumab in men with castrate-resistant prostate cancer: results from Cancer and Leukemia Group B Study 90006.Cancer. 2011 Feb 1;117(3):526-33. doi: 10.1002/cncr.25421. Epub 2010 Sep 22. Cancer. 2011. PMID: 20862750 Free PMC article. Clinical Trial.
-
Reversal of docetaxel resistance with bevacizumab and thalidomide.Clin Genitourin Cancer. 2009 Aug;7(2):E37-8. doi: 10.3816/CGC.2009.n.020. Clin Genitourin Cancer. 2009. PMID: 19692321 Free PMC article.
-
Docetaxel: a review of its use for the first-line treatment of advanced castration-resistant prostate cancer.Drugs. 2012 Jul 30;72(11):1559-77. doi: 10.2165/11209660-000000000-00000. Drugs. 2012. PMID: 22818017 Review.
-
Future directions in the treatment of androgen-independent prostate cancer.Urology. 2005 Jun;65(6 Suppl):8-12. doi: 10.1016/j.urology.2005.04.020. Urology. 2005. PMID: 15939077 Review.
Cited by
-
The lysine specific demethylase-1 (LSD1/KDM1A) regulates VEGF-A expression in prostate cancer.Mol Oncol. 2013 Jun;7(3):555-66. doi: 10.1016/j.molonc.2013.01.003. Epub 2013 Jan 19. Mol Oncol. 2013. PMID: 23384557 Free PMC article.
-
Therapeutic targeting of the prostate cancer microenvironment.Nat Rev Urol. 2010 Sep;7(9):494-509. doi: 10.1038/nrurol.2010.134. Nat Rev Urol. 2010. PMID: 20818327 Review.
-
Immunohistochemical expression of PDGFR, VEGF-C, and proteins of the mToR pathway before and after androgen deprivation therapy in prostate carcinoma: significant decrease after treatment.Target Oncol. 2014 Dec;9(4):359-66. doi: 10.1007/s11523-013-0298-1. Epub 2013 Nov 17. Target Oncol. 2014. PMID: 24243494
-
Anti-angiogenesis in prostate cancer: knocked down but not out.Asian J Androl. 2014 May-Jun;16(3):372-7. doi: 10.4103/1008-682X.125903. Asian J Androl. 2014. PMID: 24759579 Free PMC article. Review.
-
Management of metastatic castration-resistant prostate cancer: recent advances.Drugs. 2012 May 28;72(8):1011-28. doi: 10.2165/11633360-000000000-00000. Drugs. 2012. PMID: 22621691 Review.
References
-
- Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2009. CA Cancer J Clin. 2009;59:225–249. - PubMed
-
- Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–1512. - PubMed
-
- Berthold DR, Pond GR, Soban F, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: Updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242–245. - PubMed
-
- Petrylak D. Therapeutic options in androgen-independent prostate cancer: Building on docetaxel. BJU Int. 2005;96(suppl 2):41–46. - PubMed
-
- Di Lorenzo G, Figg WD, Fossa SD, et al. Combination of bevacizumab and docetaxel in docetaxel-pretreated hormone-refractory prostate cancer: A phase 2 study. Eur Urol. 2008;54:1089–1094. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

