Modeling shifts in the rate and pattern of subthalamopallidal network activity during deep brain stimulation
- PMID: 20309620
- PMCID: PMC2881193
- DOI: 10.1007/s10827-010-0225-8
Modeling shifts in the rate and pattern of subthalamopallidal network activity during deep brain stimulation
Abstract
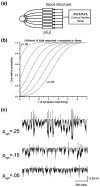
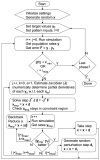
Deep brain stimulation (DBS) of the subthlamic nucleus (STN) represents an effective treatment for medically refractory Parkinson's disease; however, understanding of its effects on basal ganglia network activity remains limited. We constructed a computational model of the subthalamopallidal network, trained it to fit in vivo recordings from parkinsonian monkeys, and evaluated its response to STN DBS. The network model was created with synaptically connected single compartment biophysical models of STN and pallidal neurons, and stochastically defined inputs driven by cortical beta rhythms. A least mean square error training algorithm was developed to parameterize network connections and minimize error when compared to experimental spike and burst rates in the parkinsonian condition. The output of the trained network was then compared to experimental data not used in the training process. We found that reducing the influence of the cortical beta input on the model generated activity that agreed well with recordings from normal monkeys. Further, during STN DBS in the parkinsonian condition the simulations reproduced the reduction in GPi bursting found in existing experimental data. The model also provided the opportunity to greatly expand analysis of GPi bursting activity, generating three major predictions. First, its reduction was proportional to the volume of STN activated by DBS. Second, GPi bursting decreased in a stimulation frequency dependent manner, saturating at values consistent with clinically therapeutic DBS. And third, ablating STN neurons, reported to generate similar therapeutic outcomes as STN DBS, also reduced GPi bursting. Our theoretical analysis of stimulation induced network activity suggests that regularization of GPi firing is dependent on the volume of STN tissue activated and a threshold level of burst reduction may be necessary for therapeutic effect.
Figures








Similar articles
-
Thalamocortical relay fidelity varies across subthalamic nucleus deep brain stimulation protocols in a data-driven computational model.J Neurophysiol. 2008 Mar;99(3):1477-92. doi: 10.1152/jn.01080.2007. Epub 2008 Jan 2. J Neurophysiol. 2008. PMID: 18171706
-
Computational analysis of subthalamic nucleus and lenticular fasciculus activation during therapeutic deep brain stimulation.J Neurophysiol. 2006 Sep;96(3):1569-80. doi: 10.1152/jn.00305.2006. Epub 2006 May 31. J Neurophysiol. 2006. PMID: 16738214
-
External pallidal stimulation improves parkinsonian motor signs and modulates neuronal activity throughout the basal ganglia thalamic network.Exp Neurol. 2012 Jan;233(1):581-6. doi: 10.1016/j.expneurol.2011.09.031. Epub 2011 Oct 1. Exp Neurol. 2012. PMID: 22001773 Free PMC article.
-
Congress of Neurological Surgeons Systematic Review and Evidence-Based Guideline on Subthalamic Nucleus and Globus Pallidus Internus Deep Brain Stimulation for the Treatment of Patients With Parkinson's Disease: Executive Summary.Neurosurgery. 2018 Jun 1;82(6):753-756. doi: 10.1093/neuros/nyy037. Neurosurgery. 2018. PMID: 29538685 Free PMC article.
-
Cognition and Depression Following Deep Brain Stimulation of the Subthalamic Nucleus and Globus Pallidus Pars Internus in Parkinson's Disease: A Meta-Analysis.Neuropsychol Rev. 2015 Dec;25(4):439-54. doi: 10.1007/s11065-015-9302-0. Epub 2015 Oct 12. Neuropsychol Rev. 2015. PMID: 26459361 Review.
Cited by
-
Generic dynamic causal modelling: An illustrative application to Parkinson's disease.Neuroimage. 2018 Nov 1;181:818-830. doi: 10.1016/j.neuroimage.2018.08.039. Epub 2018 Aug 18. Neuroimage. 2018. PMID: 30130648 Free PMC article.
-
Self-Tuning Deep Brain Stimulation Controller for Suppression of Beta Oscillations: Analytical Derivation and Numerical Validation.Front Neurosci. 2020 Jun 30;14:639. doi: 10.3389/fnins.2020.00639. eCollection 2020. Front Neurosci. 2020. PMID: 32694975 Free PMC article.
-
Cortical information flow in Parkinson's disease: a composite network/field model.Front Comput Neurosci. 2013 Apr 25;7:39. doi: 10.3389/fncom.2013.00039. eCollection 2013. Front Comput Neurosci. 2013. PMID: 23630492 Free PMC article.
-
Mathematical insights into the effects of levodopa.Front Integr Neurosci. 2012 Jul 4;6:21. doi: 10.3389/fnint.2012.00021. eCollection 2012. Front Integr Neurosci. 2012. PMID: 22783173 Free PMC article.
-
Point process modeling reveals anatomical non-uniform distribution across the subthalamic nucleus in Parkinson's disease.Annu Int Conf IEEE Eng Med Biol Soc. 2012;2012:2539-42. doi: 10.1109/EMBC.2012.6346481. Annu Int Conf IEEE Eng Med Biol Soc. 2012. PMID: 23366442 Free PMC article. Clinical Trial.
References
-
- Alvarez L, Macias R, Pavon N, Lopez G, Rodriguez-Oroz MC, Rodriguez R, et al. Therapeutic efficacy of unilateral subthalamotomy in Parkinson’s disease: results in 89 patients followed for up to 36 months. Journal of Neurology, Neurosurgery and Psychiatry. 2009;80:979–985. - PubMed
-
- Bekar L, Libionka W, Tian GF, Xu Q, Torres A, Wang X, et al. Adenosine is crucial for deep brain stimulation-mediated attenuation of tremor. Nature Medicine. 2008;14:75–80. - PubMed
-
- Bergman H, Wichmann T, Karmon B, DeLong MR. The primate subthalamic nucleus. II. Neuronal activity in the MPTP model of parkinsonism. Journal of Neurophysiology. 1994;72:507–520. - PubMed
-
- Bevan MD, Magill PJ, Terman D, Bolam JP, Wilson CJ. Move to the rhythm: oscillations in the subthalamic nucleus-external globus pallidus network. Trends in Neurosciences. 2002;25(10):525–31. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

