Cell permeabilization by poliovirus 2B viroporin triggers bystander permeabilization in neighbouring cells through a mechanism involving gap junctions
- PMID: 20331640
- PMCID: PMC7162288
- DOI: 10.1111/j.1462-5822.2010.01460.x
Cell permeabilization by poliovirus 2B viroporin triggers bystander permeabilization in neighbouring cells through a mechanism involving gap junctions
Abstract
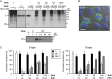
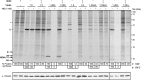
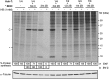
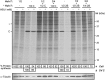
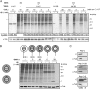
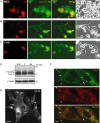
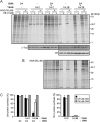
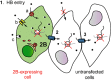
Poliovirus 2B protein is a well-known viroporin implicated in plasma membrane permeabilization to ions and low-molecular-weight compounds during infection. Translation in mammalian cells expressing 2B protein is inhibited by hygromycin B (HB) but remains unaffected in mock cells, which are not permeable to the inhibitor. Here we describe a previously unreported bystander effect in which healthy baby hamster kidney (BHK) cells become sensitive to HB when co-cultured with a low proportion of cells expressing poliovirus 2B. Viroporins E from mouse hepatitis virus, 6K from Sindbis virus and NS4A protein from hepatitis C virus were also able to permeabilize neighbouring cells to different extents. Expression of 2B induced permeabilization of neighbouring cell lines other than BHK. We found that gap junctions are responsible mediating the observed bystander permeabilization. Gap junctional communication was confirmed in 2B-expressing co-cultures by fluorescent dye transfer. Moreover, the presence of connexin 43 was confirmed in both mock and 2B-transfected cells. Finally, inhibition of HB entry to neighbouring cells was observed with 18alpha-glycyrrhethinic acid, an inhibitor of gap junctions. Taken together, these findings support a mechanism involving gap junctional intercellular communication in the bystander permeabilization effect observed in healthy cells co-cultured with poliovirus 2B-expressing cells.
Figures
References
-
- Agirre, A. , Barco, A. , Carrasco, L. , and Nieva, J.L. (2002) Viroporin‐mediated membrane permeabilization. Pore formation by nonstructural poliovirus 2B protein. J Biol Chem 277: 40434–40441. - PubMed
-
- Aldabe, R. , and Carrasco, L. (1995) Induction of membrane proliferation by poliovirus proteins 2C and 2BC. Biochem Biophys Res Commun 206: 64–76. - PubMed
-
- Aldabe, R. , Barco, A. , and Carrasco, L. (1996) Membrane permeabilization by poliovirus proteins 2B and 2 BC. J Biol Chem 271: 23134–23137. - PubMed
-
- Asklund, T. , Appelskog, I.B. , Ammerpohl, O. , Langmoen, I.A. , Dilber, M.S. , Aints, A. , et al (2003) Gap junction‐mediated bystander effect in primary cultures of human malignant gliomas with recombinant expression of the HSVtk gene. Exp Cell Res 284: 185–195. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

