Structure and functional characterization of Vibrio parahaemolyticus thermostable direct hemolysin
- PMID: 20335168
- PMCID: PMC2871494
- DOI: 10.1074/jbc.M109.074526
Structure and functional characterization of Vibrio parahaemolyticus thermostable direct hemolysin
Abstract
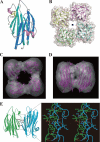
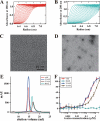
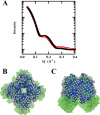
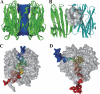
Thermostable direct hemolysin (TDH) is a major virulence factor of Vibrio parahaemolyticus that causes pandemic foodborne enterocolitis mediated by seafood. TDH exists as a tetramer in solution, and it possesses extreme hemolytic activity. Here, we present the crystal structure of the TDH tetramer at 1.5 A resolution. The TDH tetramer forms a central pore with dimensions of 23 A in diameter and approximately 50 A in depth. Pi-cation interactions between protomers comprising the tetramer were indispensable for hemolytic activity of TDH. The N-terminal region was intrinsically disordered outside of the pore. Molecular dynamic simulations suggested that water molecules permeate freely through the central and side channel pores. Electron micrographs showed that tetrameric TDH attached to liposomes, and some of the tetramer associated with liposome via one protomer. These findings imply a novel membrane attachment mechanism by a soluble tetrameric pore-forming toxin.
Figures
Similar articles
-
Relationship between heat-induced fibrillogenicity and hemolytic activity of thermostable direct hemolysin and a related hemolysin of Vibrio parahaemolyticus.FEMS Microbiol Lett. 2011 May;318(1):10-7. doi: 10.1111/j.1574-6968.2011.02233.x. Epub 2011 Mar 2. FEMS Microbiol Lett. 2011. PMID: 21291495
-
Disulphide bond restrains the C-terminal region of thermostable direct hemolysin during folding to promote oligomerization.Biochem J. 2017 Jan 15;474(2):317-331. doi: 10.1042/BCJ20160728. Epub 2016 Oct 26. Biochem J. 2017. PMID: 27784764
-
N-Terminal Region of Vibrio parahemolyticus Thermostable Direct Hemolysin Regulates the Membrane-Damaging Action of the Toxin.Biochemistry. 2020 Feb 4;59(4):605-614. doi: 10.1021/acs.biochem.9b00937. Epub 2019 Dec 20. Biochemistry. 2020. PMID: 31808340
-
Structure, function and regulation of the thermostable direct hemolysin (TDH) in pandemic Vibrio parahaemolyticus.Microb Pathog. 2018 Oct;123:242-245. doi: 10.1016/j.micpath.2018.07.021. Epub 2018 Jul 19. Microb Pathog. 2018. PMID: 30031890 Review.
-
Ecological fitness and virulence features of Vibrio parahaemolyticus in estuarine environments.Appl Microbiol Biotechnol. 2017 Mar;101(5):1781-1794. doi: 10.1007/s00253-017-8096-9. Epub 2017 Jan 31. Appl Microbiol Biotechnol. 2017. PMID: 28144705 Review.
Cited by
-
Risk Reduction Assessment of Vibrio parahaemolyticus on Shrimp by a Chinese Eating Habit.Int J Environ Res Public Health. 2022 Dec 25;20(1):317. doi: 10.3390/ijerph20010317. Int J Environ Res Public Health. 2022. PMID: 36612642 Free PMC article.
-
Current Perspective on the Membrane-Damaging Action of Thermostable Direct Hemolysin, an Atypical Bacterial Pore-forming Toxin.Front Mol Biosci. 2021 Jul 23;8:717147. doi: 10.3389/fmolb.2021.717147. eCollection 2021. Front Mol Biosci. 2021. PMID: 34368235 Free PMC article. Review.
-
Occurrence, virulence, and AMR profile of Vibrio parahaemolyticus isolated from shellfish growing areas located along the south-west coast of India.J Water Health. 2024 Sep;22(9):1594-1605. doi: 10.2166/wh.2024.338. Epub 2024 Aug 6. J Water Health. 2024. PMID: 39340373
-
Site-directed mutations of thermostable direct hemolysin from Grimontia hollisae alter its arrhenius effect and biophysical properties.Int J Biol Sci. 2011 Mar 31;7(3):333-46. doi: 10.7150/ijbs.7.333. Int J Biol Sci. 2011. PMID: 21494434 Free PMC article.
-
Structural and biophysical characterization of Bacillus thuringiensis insecticidal proteins Cry34Ab1 and Cry35Ab1.PLoS One. 2014 Nov 12;9(11):e112555. doi: 10.1371/journal.pone.0112555. eCollection 2014. PLoS One. 2014. PMID: 25390338 Free PMC article.
References
-
- Yamaichi Y., Iida T., Park K. S., Yamamoto K., Honda T. (1999) Mol. Microbiol. 31, 1513–1521 - PubMed
-
- Makino K., Oshima K., Kurokawa K., Yokoyama K., Uda T., Tagomori K., Iijima Y., Najima M., Nakano M., Yamashita A., Kubota Y., Kimura S., Yasunaga T., Honda T., Shinagawa H., Hattori M., Iida T. (2003) Lancet 361, 743–749 - PubMed
-
- Iida T., Honda T. (1997) Toxin Rev. 16, 215–227
-
- Honda T., Iida T., Akeda A., Kodama T. (2008) Microbe 3, 462–466
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

