Abnormal hair follicle development and altered cell fate of follicular keratinocytes in transgenic mice expressing DeltaNp63alpha
- PMID: 20335364
- PMCID: PMC2853845
- DOI: 10.1242/dev.045427
Abnormal hair follicle development and altered cell fate of follicular keratinocytes in transgenic mice expressing DeltaNp63alpha
Erratum in
- Development. 2010 May;137(10):1775
Abstract
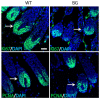
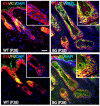
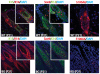
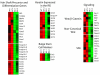
The transcription factor p63 plays an essential role in epidermal morphogenesis. Animals lacking p63 fail to form many ectodermal organs, including the skin and hair follicles. Although the indispensable role of p63 in stratified epithelial skin development is well established, relatively little is known about this transcriptional regulator in directing hair follicle morphogenesis. Here, using specific antibodies, we have established the expression pattern of DeltaNp63 in hair follicle development and cycling. DeltaNp63 is expressed in the developing hair placode, whereas in mature hair its expression is restricted to the outer root sheath (ORS), matrix cells and to the stem cells of the hair follicle bulge. To investigate the role of DeltaNp63 in hair follicle morphogenesis and cycling, we have utilized a Tet-inducible mouse model system with targeted expression of this isoform to the ORS of the hair follicle. DeltaNp63 transgenic animals display dramatic defects in hair follicle development and cycling, eventually leading to severe hair loss. Strikingly, expression of DeltaNp63 leads to a switch in cell fate of hair follicle keratinocytes, causing them to adopt an interfollicular epidermal (IFE) cell identity. Moreover, DeltaNp63 transgenic animals exhibit a depleted hair follicle stem-cell niche, which further contributes to the overall cycling defects observed in the mutant animals. Finally, global transcriptome analysis of transgenic skin identified altered expression levels of crucial mediators of hair morphogenesis, including key members of the Wnt/beta-catenin signaling pathway, which, in part, account for these effects. Our data provide evidence supporting a role for DeltaNp63alpha in actively suppressing hair follicle differentiation and directing IFE cell lineage commitment.
Figures




References
-
- Andl T., Reddy S. T., Gaddapara T., Millar S. E. (2002). WNT signals are required for the initiation of hair follicle development. Dev. Cell 2, 643-653 - PubMed
-
- Barbieri C. E., Pietenpol J. A. (2006). p63 and epithelial biology. Exp. Cell Res. 312, 695-706 - PubMed
-
- Blanpain C., Fuchs E. (2007). p63: revving up epithelial stem-cell potential. Nat. Cell Biol. 9, 731-733 - PubMed
-
- Callahan C. A., Oro A. E. (2001). Monstrous attempts at adnexogenesis: regulating hair follicle progenitors through Sonic hedgehog signaling. Curr. Opin. Genet. Dev. 11, 541-546 - PubMed
-
- Candi E., Rufini A., Terrinoni A., Dinsdale D., Ranalli M., Paradisi A., De Laurenzi V., Spagnoli L. G., Catani M. V., Ramadan S., et al. (2006). Differential roles of p63 isoforms in epidermal development: selective genetic complementation in p63 null mice. Cell Death Differ. 13, 1037-1047 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

