TASK channels contribute to the K+-dominated leak current regulating respiratory rhythm generation in vitro
- PMID: 20335463
- PMCID: PMC2950010
- DOI: 10.1523/JNEUROSCI.4017-09.2010
TASK channels contribute to the K+-dominated leak current regulating respiratory rhythm generation in vitro
Abstract
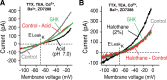
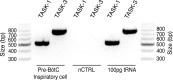
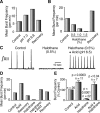
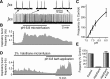
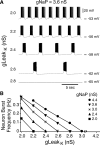
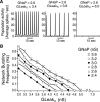
Leak channels regulate neuronal activity and excitability. Determining which leak channels exist in neurons and how they control electrophysiological behavior is fundamental. Here we investigated TASK channels, members of the two-pore domain K(+) channel family, as a component of the K(+)-dominated leak conductance that controls and modulates rhythm generation at cellular and network levels in the mammalian pre-Bötzinger complex (pre-BötC), an excitatory network of neurons in the medulla critically involved in respiratory rhythmogenesis. By voltage-clamp analyses of pre-BötC neuronal current-voltage (I-V) relations in neonatal rat medullary slices in vitro, we demonstrated that pre-BötC inspiratory neurons have a weakly outward-rectifying total leak conductance with reversal potential that was depolarized by approximately 4 mV from the K(+) equilibrium potential, indicating that background K(+) channels are dominant contributors to leak. This K(+) channel component had I-V relations described by constant field theory, and the conductance was reduced by acid and was augmented by the volatile anesthetic halothane, which are all hallmarks of TASK. We established by single-cell RT-PCR that pre-BötC inspiratory neurons express TASK-1 and in some cases also TASK-3 mRNA. Furthermore, acid depolarized and augmented bursting frequency of pre-BötC inspiratory neurons with intrinsic bursting properties. Microinfusion of acidified solutions into the rhythmically active pre-BötC network increased network bursting frequency, halothane decreased bursting frequency, and acid reversed the depressant effects of halothane, consistent with modulation of network activity by TASK channels. We conclude that TASK-like channels play a major functional role in chemosensory modulation of respiratory rhythm generation in the pre-Bötzinger complex in vitro.
Figures



References
-
- Bayliss DA, Siros JE, Talley EM. The TASK family: two-pore-domain background K+ channels. Mol Interven. 2003;3:205–219. - PubMed
-
- Burdakov D, Jensen LT, Alexopoulos H, Williams RH, Fearon IM, O'Kelly I, Gerasimenko O, Fugger L, Verkhratsky A. Tandem-pore K+ channels mediate inhibition of orexin neurons by glucose. Neuron. 2006;50:711–722. - PubMed
-
- Butera RJ, Jr, Rinzel J, Smith JC. Models of respiratory rhythm generation in the pre-Botzinger complex. I. Bursting pacemaker neurons. J Neurophysiol. 1999a;82:382–397. - PubMed
-
- Butera RJ, Jr, Rinzel J, Smith JC. Models of respiratory rhythm generation in the pre-Botzinger complex. II. Populations of coupled pacemaker neurons. J Neurophysiol. 1999b;82:398–415. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
