Regulation of immune cell development through soluble inositol-1,3,4,5-tetrakisphosphate
- PMID: 20336153
- PMCID: PMC2922113
- DOI: 10.1038/nri2745
Regulation of immune cell development through soluble inositol-1,3,4,5-tetrakisphosphate
Abstract
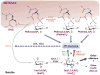
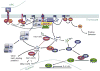
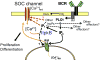
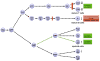
The membrane lipid phosphatidylinositol-3,4,5-trisphosphate (PtdInsP(3)) regulates membrane receptor signalling in many cells, including immunoreceptor signalling. Here, we review recent data that have indicated essential roles for the soluble PtdInsP(3) analogue inositol-1,3,4,5-tetrakisphosphate (InsP(4)) in T cell, B cell and neutrophil development and function. Decreased InsP(4) production in leukocytes causes immunodeficiency in mice and might contribute to inflammatory vasculitis in Kawasaki disease in humans. InsP(4)-producing kinases could therefore provide attractive drug targets for inflammatory and infectious diseases.
Figures

References
-
- York JD. Regulation of nuclear processes by inositol polyphosphates. Biochim Biophys Acta. 2006;1761:552–559. - PubMed
-
- Michell RH, Heath VL, Lemmon MA, Dove SK. Phosphatidylinositol 3,5-bisphosphate: metabolism and cellular functions. Trends Biochem Sci. 2006;31:52–63. This notable review proposes a unified standard nomenclature for inositol phosphates. - PubMed
-
- Streb H, Irvine RF, Berridge MJ, Schulz I. Release of Ca2+ from a nonmitochondrial intracellular store in pancreatic acinar cells by inositol-1,4,5-trisphosphate. Nature. 1983;306:67–69. This seminal publication first described that Ins(1,4,5)P3 acts as a second messenger which mediates Ca2+-release in mammalian cells. - PubMed
-
- Skwarek LC, Boulianne GL. Great expectations for PIP: phosphoinositides as regulators of signaling during development and disease. Dev Cell. 2009;16:12–20. - PubMed
-
- Fruman DA, Bismuth G. Fine tuning the immune response with PI3K. Immunol Rev. 2009;228:253–272. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

