2-MeS-beta,gamma-CCl2-ATP is a potent agent for reducing intraocular pressure
- PMID: 20337495
- PMCID: PMC4358770
- DOI: 10.1021/jm100030u
2-MeS-beta,gamma-CCl2-ATP is a potent agent for reducing intraocular pressure
Abstract
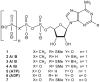
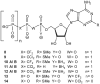
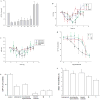
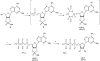
Extracellular nucleotides can modify the production or drainage of the aqueous humor via activation of P2 receptors and therefore affect the intraocular pressure (IOP). We have synthesized slowly hydrolyzable nucleoside di- and triphosphate analogues, 1, and 8-14. Analogues 8-14 were completely resistant to hydrolysis by alkaline phosphatase over 30 min at 37 degrees C. In human blood serum, analogues 8-14 exhibited high stability, e.g., analogues 9 and 10-14 were only 15% and 0% degraded after 24 h, respectively. Moreover, analogues 8-14 were highly stable at pH 1.4 (t(1/2) 1 h-30 days). Analogues 8-14 were agonists of the P2Y(1) receptor (EC(50) 0.57-9.54 muM). Ocular administration of most analogues into rabbits reduced IOP, e.g., analogue 9 reduced IOP by 32% (EC(50) 95.5 nM). Analogue 9 was more effective at reducing IOP than several common glaucoma drugs and represents a promising alternative to timolol maleate, which cannot be used for the treatment of patients suffering from asthma or cardiac problems.
Figures




References
-
- Williams M, Jarvis MF. Purinergic and pyrimidinergic receptors as potential drug targets. Biochem Pharmacol. 2000;59:1173–1185. - PubMed
-
- Guile SD, Ince F, Ingall AH, Kindon ND, Meghani P, Mortimore MP. The medicinal chemistry of the P2 receptor family. Prog Med Chem. 2001;38:115–187. - PubMed
-
- Fischer B. Therapeutic applications of ATP-(P2)-receptors agonists and antagonists. Expert Opin Ther Pat. 1999;9:385–399.
-
- Hillmann P, Ko GY, Spinrath A, Raulf A, von Kugelgen I, Wolff SC, Nicholas RA, Kostenis E, Holtje HD, Muller CE. Key Determinants of nucleotide-activated G protein-coupled P2Y2 receptor function revealed by chemical and pharmacological experiments, mutagenesis and homology modeling. J Med Chem. 2009;52:2762–2775. - PubMed
-
- Burnstock G, Verkhratsky A. Evolutionary origins of the purinergic signalling system. Acta Physiol. 2009;195:415–447. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Miscellaneous

