Lack of SIRT1 (Mammalian Sirtuin 1) activity leads to liver steatosis in the SIRT1+/- mice: a role of lipid mobilization and inflammation
- PMID: 20339025
- PMCID: PMC2875813
- DOI: 10.1210/en.2009-1013
Lack of SIRT1 (Mammalian Sirtuin 1) activity leads to liver steatosis in the SIRT1+/- mice: a role of lipid mobilization and inflammation
Abstract
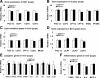
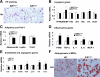
Mammalian sirtuin 1 (SIRT1) may control fatty acid homeostasis in liver. However, this possibility and underlying mechanism remain to be established. In this study, we addressed the issues by examining the metabolic phenotypes of SIRT1 heterozygous knockout (SIRT1(+/-)) mice. The study was conducted in the mice on three different diets including a low-fat diet (5% fat wt/wt), mediate-fat diet (11% fat wt/wt), and high-fat diet (HFD, 36% fat wt/wt). On low-fat diet, the mice did not exhibit any abnormality. On mediate-fat diet, the mice exhibited a significant increase in hepatic steatosis with elevated liver/body ratio, liver size, liver lipid (triglyceride, glycerol, and cholesterol) content, and liver inflammation. The hepatic steatosis was deteriorated in the mice by HFD. In the liver, lipogenesis was increased, fat export was reduced, and beta-oxidation was not significantly changed. Body weight and fat content were increased in response to the dietary fat. Fat was mainly increased in sc adipose tissue and liver. Inflammation was also elevated in epididymal fat. Whole body energy expenditure and substrate utilization were reduced. Food intake, locomotor activity, and fat absorption were not changed. These data suggest that a reduction in the SIRT1 activity increases the risk of fatty liver in response to dietary fat. The liver steatosis may be a result of increased lipogenesis and reduced liver fat export. The inflammation may contribute to the pathogenesis of hepatic steatosis as well. A reduction in lipid mobilization may contribute to the hepatic steatosis and low energy expenditure.
Figures




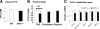
References
-
- Imai S, Armstrong CM, Kaeberlein M, Guarente L 2000 Transcriptional silencing and longevity protein Sir2 is an NAD-dependent histone deacetylase. Nature 403:795–800 - PubMed
-
- Lin SJ, Defossez PA, Guarente L 2000 Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science 289:2126–2128 - PubMed
-
- Lombard DB, Chua KF, Mostoslavsky R, Franco S, Gostissa M, Alt FW 2005 DNA repair, genome stability, and aging. Cell 120:497–512 - PubMed
-
- Longo VD, Kennedy BK 2006 Sirtuins in aging and age-related disease. Cell 126:257–268 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

