Predicting clinical scores from magnetic resonance scans in Alzheimer's disease
- PMID: 20347044
- PMCID: PMC2871976
- DOI: 10.1016/j.neuroimage.2010.03.051
Predicting clinical scores from magnetic resonance scans in Alzheimer's disease
Abstract
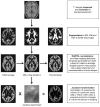
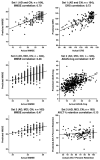
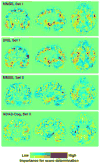
Machine learning and pattern recognition methods have been used to diagnose Alzheimer's disease (AD) and mild cognitive impairment (MCI) from individual MRI scans. Another application of such methods is to predict clinical scores from individual scans. Using relevance vector regression (RVR), we predicted individuals' performances on established tests from their MRI T1 weighted image in two independent data sets. From Mayo Clinic, 73 probable AD patients and 91 cognitively normal (CN) controls completed the Mini-Mental State Examination (MMSE), Dementia Rating Scale (DRS), and Auditory Verbal Learning Test (AVLT) within 3months of their scan. Baseline MRI's from the Alzheimer's disease Neuroimaging Initiative (ADNI) comprised the other data set; 113 AD, 351 MCI, and 122 CN subjects completed the MMSE and Alzheimer's Disease Assessment Scale-Cognitive subtest (ADAS-cog) and 39 AD, 92 MCI, and 32 CN ADNI subjects completed MMSE, ADAS-cog, and AVLT. Predicted and actual clinical scores were highly correlated for the MMSE, DRS, and ADAS-cog tests (P<0.0001). Training with one data set and testing with another demonstrated stability between data sets. DRS, MMSE, and ADAS-Cog correlated better than AVLT with whole brain grey matter changes associated with AD. This result underscores their utility for screening and tracking disease. RVR offers a novel way to measure interactions between structural changes and neuropsychological tests beyond that of univariate methods. In clinical practice, we envision using RVR to aid in diagnosis and predict clinical outcome.
Copyright 2010 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors have no conflict of interest or financial involvement with this manuscript.
Figures

References
-
- American Psychiatric Association. Diagnostic and statistical manual of mental disorders. Washington, D.C.: American Psychiatric Press; 1987.
-
- Apostolova LG, Lu PH, Rogers S, Dutton RA, Hayashi KM, Toga AW, Cummings JL, Thompson PM. 3D mapping of mini-mental state examination performance in clinical and preclinical Alzheimer disease. Alzheimer Dis Assoc Disord. 2006;20:224–231. - PubMed
-
- Ashburner J. A fast diffeomorphic image registration algorithm. Neuroimage. 2007;38:95–113. - PubMed
-
- Ashburner J, Friston KJ. Voxel-based morphometry--the methods. Neuroimage. 2000;11:805–821. - PubMed
-
- Ashburner J, Friston KJ. Unified segmentation. Neuroimage. 2005;26:839–851. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

