Phase I clinical trials in 85 patients with gynecologic cancer: the M. D. Anderson Cancer Center experience
- PMID: 20347123
- PMCID: PMC2882061
- DOI: 10.1016/j.ygyno.2010.02.008
Phase I clinical trials in 85 patients with gynecologic cancer: the M. D. Anderson Cancer Center experience
Abstract
Objective: Disseminated gynecologic cancers are usually fatal due to chemoresistance. Recently, rationally developed, targeted agents are entering the early clinical trials setting. We assessed patients with metastatic gynecologic cancers in a dedicated phase I clinical trials clinic in order to determine their outcome.
Methods: We reviewed records for 89 consecutive patients with gynecologic cancers referred to the Phase I Clinical Trials Program, 85 (96%) of whom were treated on > or = 1 trial.
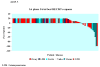
Results: Cancer diagnoses were ovarian (N=43), uterine (N=19), cervix (N=17), and other. Median age was 58 years; median number of prior cytotoxic regimens, five. Two patients (2.4%) achieved a CR; four (4.7%), a PR; and eight (9.4%), SD > or = 6 months (total CR/PR/SD > or = 6 months=16.5%) for the first phase I trial. Twenty-five patients enrolled on a second trial and three, on a third (N=113 trials total). Combining response data for all trials, of the 85 patients, two achieved CR (2.4%), nine achieved PR (10.6%), and 12 (14%) had SD for > or = 6 months. One-year survival was 30% (95% CI, 21% to 44%). There was no difference in time-to-treatment failure (TTF) on phase I versus the patient's last standard treatment.
Conclusion: Twenty-three of 85 patients (27%) with advanced, heavily pretreated, gynecologic cancers achieved CR/PR/SD > or = 6 months on a phase I trial, and overall TTF on phase I was comparable to that of last conventional therapy, suggesting that participation in a phase I trial is a reasonable option for these patients.
Copyright 2010 Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures



References
-
- Lu KH. Management of early-stage endometrial cancer. Semin Oncol. 2009;36(2):137–44. - PubMed
-
- Lee CM, et al. Frequency and effect of adjuvant radiation therapy among women with stage I endometrial adenocarcinoma. Jama. 2006;295(4):389–97. - PubMed
-
- Warwick J, et al. Defining the surgical management of suspected early-stage ovarian cancer by estimating patient numbers through alternative management strategies. Bjog. 2009;116(9):1225–41. - PubMed
-
- Armstrong DK. Relapsed ovarian cancer: challenges and management strategies for a chronic disease. Oncologist. 2002;7 5:20–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

