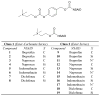
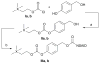
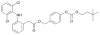Peripheral site acetylcholinesterase inhibitors targeting both inflammation and cholinergic dysfunction
- PMID: 20347302
- PMCID: PMC2864113
- DOI: 10.1016/j.bmcl.2010.02.102
Peripheral site acetylcholinesterase inhibitors targeting both inflammation and cholinergic dysfunction
Abstract
The design and study of two classes of noncompetitive acetylcholinesterase inhibitors (AChEIs) which also function as NSAID prodrugs are reported. The most potent AChEIs disclosed contain an aromatic alkyl-aryl linker between an NSAID and a lipophilic choline mimic and they inhibit acetylcholinesterase (AChE) in the submicromolar range. These agents have the therapeutic potential to dually target inflammation by releasing an NSAID in vivo and activating the cholinergic anti-inflammatory pathway via cholinergic up-regulation.
2010 Elsevier Ltd. All rights reserved.
Figures
Similar articles
-
One-pot synthesis of tetrazole-1,2,5,6-tetrahydronicotinonitriles and cholinesterase inhibition: Probing the plausible reaction mechanism via computational studies.Bioorg Chem. 2016 Apr;65:38-47. doi: 10.1016/j.bioorg.2016.01.004. Epub 2016 Jan 21. Bioorg Chem. 2016. PMID: 26851737
-
Rational design of carbamate-based dual binding site and central AChE inhibitors by a "biooxidisable" prodrug approach: Synthesis, in vitro evaluation and docking studies.Eur J Med Chem. 2018 Jul 15;155:171-182. doi: 10.1016/j.ejmech.2018.05.057. Epub 2018 Jun 1. Eur J Med Chem. 2018. PMID: 29886321
-
Investigation of anticholinergic and non-steroidal anti-inflammatory prodrugs which reduce chemically induced skin inflammation.J Appl Toxicol. 2012 Feb;32(2):135-41. doi: 10.1002/jat.1645. Epub 2011 Feb 11. J Appl Toxicol. 2012. PMID: 21319177 Free PMC article.
-
Rational design of reversible acetylcholinesterase inhibitors.Mini Rev Med Chem. 2002 Feb;2(1):27-36. doi: 10.2174/1389557023406494. Mini Rev Med Chem. 2002. PMID: 12369955 Review.
-
Peripheral and dual binding site acetylcholinesterase inhibitors: implications in treatment of Alzheimer's disease.Mini Rev Med Chem. 2001 Sep;1(3):267-72. doi: 10.2174/1389557013406864. Mini Rev Med Chem. 2001. PMID: 12369973 Review.
Cited by
-
Multi-inhibitor prodrug constructs for simultaneous delivery of anti-inflammatory agents to mustard-induced skin injury.Ann N Y Acad Sci. 2016 Aug;1378(1):174-179. doi: 10.1111/nyas.13177. Epub 2016 Aug 9. Ann N Y Acad Sci. 2016. PMID: 27505078 Free PMC article. Review.
-
Depilatory double-disc mouse model for evaluation of vesicant dermal injury pharmacotherapy countermeasures.Animal Model Exp Med. 2023 Feb;6(1):57-65. doi: 10.1002/ame2.12304. Animal Model Exp Med. 2023. PMID: 36872306 Free PMC article.
-
Back-scattering interferometry: an ultrasensitive method for the unperturbed detection of acetylcholinesterase-inhibitor interactions.Angew Chem Int Ed Engl. 2012 Oct 29;51(44):11126-30. doi: 10.1002/anie.201203640. Epub 2012 Oct 4. Angew Chem Int Ed Engl. 2012. PMID: 23037915 Free PMC article.
-
Pain Chemogenomics Knowledgebase (Pain-CKB) for Systems Pharmacology Target Mapping and Physiologically Based Pharmacokinetic Modeling Investigation of Opioid Drug-Drug Interactions.ACS Chem Neurosci. 2020 Oct 21;11(20):3245-3258. doi: 10.1021/acschemneuro.0c00372. Epub 2020 Oct 7. ACS Chem Neurosci. 2020. PMID: 32966035 Free PMC article.
-
Mitigation of nitrogen mustard mediated skin injury by a novel indomethacin bifunctional prodrug.Exp Mol Pathol. 2016 Jun;100(3):522-31. doi: 10.1016/j.yexmp.2016.05.008. Epub 2016 May 14. Exp Mol Pathol. 2016. PMID: 27189522 Free PMC article.
References
-
- Özgüney I. Expert Opin Pharmacother. 2008;9:1805–1816. - PubMed
-
- Cannon J. Pharmacology for Chemists. 2. Oxford University Press; New York: Concepts of Pharmacology; pp. 13–17.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information