Hypertrophic cardiomyopathy
- PMID: 20347784
- PMCID: PMC3031113
- DOI: 10.1016/j.hfc.2009.12.001
Hypertrophic cardiomyopathy
Abstract
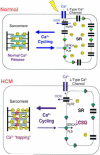
Important insights into the molecular basis of hypertrophic cardiomyopathy and related diseases have been gained by studying families with inherited cardiac hypertrophy. Integrated clinical and genetic investigations have demonstrated that different genetic defects can give rise to the common phenotype of cardiac hypertrophy. Diverse pathways have been identified, implicating perturbations in force generation, force transmission, intracellular calcium homeostasis, myocardial energetics, and cardiac metabolism in causing disease. Although not fully elucidated, the fundamental mechanisms linking gene mutations to clinical disease are being characterized. Further advances will allow a better understanding of pathogenesis, diagnosis, and treatment, not just of relatively rare inherited cardiomyopathies, but potentially also of relevance to more common acquired forms of hypertrophic remodeling.
Copyright (c) 2010 Elsevier Inc. All rights reserved.
Figures









References
-
- Adabag AS, Maron BJ, Appelbaum E, et al. Occurrence and frequency of arrhythmias in hypertrophic cardiomyopathy in relation to delayed enhancement on cardiovascular magnetic resonance. J Am Coll Cardiol. 2008;51:1369. - PubMed
-
- Ahmad F, Seidman JG, Seidman CE. The genetic basis for cardiac remodeling. Annu Rev Genomics Hum Genet. 2005;6:185. - PubMed
-
- Arad M, Maron BJ, Gorham JM, et al. Glycogen storage diseases presenting as hypertrophic cardiomyopathy. N Engl J Med. 2005;352:362. - PubMed
-
- Ashrafian H, Watkins H. Reviews of translational medicine and genomics in cardiovascular disease: new disease taxonomy and therapeutic implications cardiomyopathies: therapeutics based on molecular phenotype. J Am Coll Cardiol. 2007;49:1251. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

