Tunnel propagation following defibrillation with ICD shocks: hidden postshock activations in the left ventricular wall underlie isoelectric window
- PMID: 20348028
- PMCID: PMC2908526
- DOI: 10.1016/j.hrthm.2010.03.026
Tunnel propagation following defibrillation with ICD shocks: hidden postshock activations in the left ventricular wall underlie isoelectric window
Abstract
Background: After near-defibrillation threshold (DFT) shocks from an implantable cardioverter-defibrillator (ICD), the first postshock activation that leads to defibrillation failure arises focally after an isoelectric window (IW). The mechanisms underlying the IW remain incompletely understood.
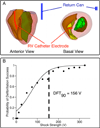
Objective: The goal of this study was to provide mechanistic insight into the origins of postshock activations and IW after ICD shocks, and to link shock outcome to the preshock state of the ventricles. We hypothesized that the nonuniform ICD field results in the formation of an intramural excitable area (tunnel) only in the left ventricular (LV) free wall, through which both pre-existing and new shock-induced wavefronts propagate during the IW.
Methods: Simulations were conducted using a realistic three dimensional (3D) model of defibrillation in the rabbit ventricles. Biphasic ICD shocks of varying strengths were delivered to 27 different fibrillatory states.
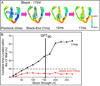
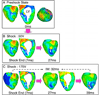
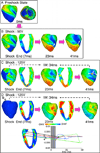
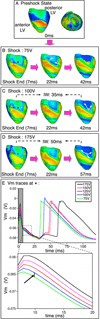
Results: After near-DFT shocks, regardless of preshock state, the main postshock excitable area was always located within LV free wall, creating an intramural tunnel. Either pre-existing fibrillatory or shock-induced wavefronts propagated during the IW (duration of up to 74 ms) in this tunnel and emerged as breakthroughs on LV epicardium. Preshock activity within the LV played a significant role in shock outcome: a large number of preshock filaments resulted in an IW associated with tunnel propagation of pre-existing rather than shock-induced wavefronts. Furthermore, shocks were more likely to succeed if the LV excitable area was smaller.
Conclusion: The LV intramural excitable area is the primary reason for near-DFT failure. Any intervention that decreases the extent of this area will improve the likelihood of defibrillation success.
Copyright 2010 Heart Rhythm Society. Published by Elsevier Inc. All rights reserved.
Figures



Comment in
-
Tunnel propagation after defibrillation: a light at the end of the tunnel.Heart Rhythm. 2010 Jul;7(7):962-3. doi: 10.1016/j.hrthm.2010.04.016. Epub 2010 Apr 20. Heart Rhythm. 2010. PMID: 20416400 No abstract available.
References
-
- Yamaguchi H, Weil M, Tang W, et al. Myocardial dysfunction after electrical defibrillation. Resuscitation. 2002;54(3):289–296. - PubMed
-
- Chattipakorn N, Fotuhi PC, Chattipakorn SC, Ideker RE. Three-dimensional mapping of earliest activation after near-threshold ventricular defibrillation shocks. J Cardiovasc Electrophysiol. 2003;14(1):65–69. - PubMed
-
- Chattipakorn N, Banville I, Gray RA, Ideker RE. Mechanism of ventricular defibrillation for near-defibrillation threshold shocks: a whole-heart optical mapping study in swine. Circulation. 2001;104(11):1313–1319. - PubMed
-
- Trayanova NA, Gray RA, Bourn DW, Eason JC. Virtual electrode-induced positive and negative graded responses: new insights into fibrillation induction and defibrillation. J Cardiovasc Electrophysiol. 2003;14(7):756–763. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

