Glutathionylation of adenine nucleotide translocase induced by carbon monoxide prevents mitochondrial membrane permeabilization and apoptosis
- PMID: 20348099
- PMCID: PMC2878049
- DOI: 10.1074/jbc.M109.065052
Glutathionylation of adenine nucleotide translocase induced by carbon monoxide prevents mitochondrial membrane permeabilization and apoptosis
Abstract
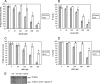
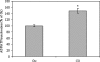
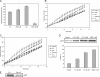
The present work demonstrates the ability of CO to prevent apoptosis in a primary culture of astrocytes. For the first time, the antiapoptotic behavior can be clearly attributed to the inhibition of mitochondrial membrane permeabilization (MMP), a key event in the intrinsic apoptotic pathway. In isolated non-synaptic mitochondria, CO partially inhibits (i) loss of potential, (ii) the opening of a nonspecific pore through the inner membrane, (iii) swelling, and (iv) cytochrome c release, which are induced by calcium, diamide, or atractyloside (a ligand of ANT). CO directly modulates ANT function by enhancing ADP/ATP exchange and prevents its pore-forming activity. Additionally, CO induces reactive oxygen species (ROS) generation, and its prevention by beta-carotene decreases CO cytoprotection in intact cells as well as in isolated mitochondria, revealing the key role of ROS. On the other hand, CO induces a slight increase in mitochondrial oxidized glutathione, which is essential for apoptosis modulation by (i) delaying astrocytic apoptosis, (ii) decreasing MMP, and (iii) enhancing ADP/ATP translocation activity of ANT. Moreover, CO and GSSG trigger ANT glutathionylation, a post-translational process regulating protein function in response to redox cellular changes. In conclusion, CO protects astrocytes from apoptosis by preventing MMP, acting on ANT (glutathionylation and inhibition of its pore activity) via a preconditioning-like process mediated by ROS and GSSG.
Figures






Similar articles
-
A possible cooperation of SOD1 and cytochrome c in mitochondria-dependent apoptosis.Free Radic Biol Med. 2006 Jan 1;40(1):173-81. doi: 10.1016/j.freeradbiomed.2005.09.037. Epub 2005 Oct 21. Free Radic Biol Med. 2006. PMID: 16337891
-
Carbon monoxide prevents hepatic mitochondrial membrane permeabilization.BMC Cell Biol. 2011 Mar 9;12:10. doi: 10.1186/1471-2121-12-10. BMC Cell Biol. 2011. PMID: 21388535 Free PMC article.
-
GAPDH, a novel regulator of the pro-apoptotic mitochondrial membrane permeabilization.Oncogene. 2007 Apr 19;26(18):2606-20. doi: 10.1038/sj.onc.1210074. Epub 2006 Oct 30. Oncogene. 2007. PMID: 17072346
-
Permeabilization of the mitochondrial inner membrane during apoptosis: impact of the adenine nucleotide translocator.Cell Death Differ. 2000 Dec;7(12):1146-54. doi: 10.1038/sj.cdd.4400778. Cell Death Differ. 2000. PMID: 11175251 Review.
-
The adenine nucleotide translocase 2, a mitochondrial target for anticancer biotherapy.Curr Drug Targets. 2011 Jun;12(6):894-901. doi: 10.2174/138945011795529047. Curr Drug Targets. 2011. PMID: 21269262 Review.
Cited by
-
Carbon monoxide modulates apoptosis by reinforcing oxidative metabolism in astrocytes: role of Bcl-2.J Biol Chem. 2012 Mar 30;287(14):10761-70. doi: 10.1074/jbc.M111.306738. Epub 2012 Feb 13. J Biol Chem. 2012. PMID: 22334654 Free PMC article.
-
An evolving understanding of the S-glutathionylation cycle in pathways of redox regulation.Free Radic Biol Med. 2018 May 20;120:204-216. doi: 10.1016/j.freeradbiomed.2018.03.038. Epub 2018 Mar 23. Free Radic Biol Med. 2018. PMID: 29578070 Free PMC article. Review.
-
Protein redox modification as a cellular defense mechanism against tissue ischemic injury.Oxid Med Cell Longev. 2014;2014:343154. doi: 10.1155/2014/343154. Epub 2014 May 5. Oxid Med Cell Longev. 2014. PMID: 24883175 Free PMC article. Review.
-
The role of gasotransmitters NO, H2S and CO in myocardial ischaemia/reperfusion injury and cardioprotection by preconditioning, postconditioning and remote conditioning.Br J Pharmacol. 2015 Mar;172(6):1587-606. doi: 10.1111/bph.12811. Epub 2014 Sep 23. Br J Pharmacol. 2015. PMID: 24923364 Free PMC article. Review.
-
The Measurement of Reversible Redox Dependent Post-translational Modifications and Their Regulation of Mitochondrial and Skeletal Muscle Function.Front Physiol. 2015 Nov 25;6:347. doi: 10.3389/fphys.2015.00347. eCollection 2015. Front Physiol. 2015. PMID: 26635632 Free PMC article. Review.
References
-
- Kirino T. (2002) J. Cereb. Blood Flow Metab. 22, 1283–1296 - PubMed
-
- Dirnagl U., Meisel A. (2008) Neuropharmacology 55, 334–344 - PubMed
-
- Wiegand F., Liao W., Busch C., Castell S., Knapp F., Lindauer U., Megow D., Meisel A., Redetzky A., Ruscher K., Trendelenburg G., Victorov I., Riepe M., Diener H. C., Dirnagl U. (1999) J. Cereb. Blood Flow Metab. 19, 1229–1237 - PubMed
-
- Kroemer G., Galluzzi L., Brenner C. (2007) Physiol. Rev. 87, 99–163 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

