The transcriptional repressor FarR is not involved in meningococcal fatty acid resistance mediated by the FarAB efflux pump and dependent on lipopolysaccharide structure
- PMID: 20348314
- PMCID: PMC2869119
- DOI: 10.1128/AEM.02833-09
The transcriptional repressor FarR is not involved in meningococcal fatty acid resistance mediated by the FarAB efflux pump and dependent on lipopolysaccharide structure
Abstract
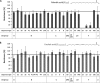
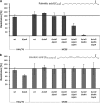
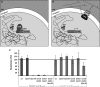
Free fatty acids are important antimicrobial substances regulating the homeostasis of colonizing bacteria on epithelial surfaces. Here, we show that meningococci express a functional farAB efflux pump, which is indispensable for fatty acid resistance. However, other than in Neisseria gonorrhoeae, the transcriptional regulator FarR is not involved in regulation of this operon in Neisseria meningitidis. We tested the susceptibility of 23 meningococcal isolates against saturated and unsaturated long-chain fatty acids, proving that meningococci are generally highly resistant, with the exception of serogroup Y strains belonging to sequence type 23. Using genetically determined lipopolysaccharide (LPS)-truncated mutant strains, we show that addition of the LPS core oligosaccharide and hexa-acylation of its membrane anchor lipid A are imperative for fatty acid resistance of meningococci. The sensitivity of the serogroup Y strains is due to naturally occurring mutations within the lpxL1 gene, which is responsible for addition of the sixth acyl chain on the LPS membrane anchor lipid A. Therefore, fatty acid resistance in meningococci is provided by both the active efflux pump FarAB and by the natural permeability barrier of the Gram-negative outer membrane. The transcriptional regulator FarR is not implicated in fatty acid resistance in meningococci, possibly giving rise to a constitutively active FarAB efflux pump system and thus revealing diverse mechanisms of niche adaptation in the two closely related Neisseria species.
Figures

Similar articles
-
Integration Host Factor is required for FarR repression of the farAB-encoded efflux pump of Neisseria gonorrhoeae.Mol Microbiol. 2006 Jun;60(6):1381-400. doi: 10.1111/j.1365-2958.2006.05185.x. Mol Microbiol. 2006. PMID: 16796676
-
FarR regulates the farAB-encoded efflux pump of Neisseria gonorrhoeae via an MtrR regulatory mechanism.J Bacteriol. 2003 Dec;185(24):7145-52. doi: 10.1128/JB.185.24.7145-7152.2003. J Bacteriol. 2003. PMID: 14645274 Free PMC article.
-
Cationic antimicrobial peptide resistance in Neisseria meningitidis.J Bacteriol. 2005 Aug;187(15):5387-96. doi: 10.1128/JB.187.15.5387-5396.2005. J Bacteriol. 2005. PMID: 16030233 Free PMC article.
-
Genetic organization and regulation of antimicrobial efflux systems possessed by Neisseria gonorrhoeae and Neisseria meningitidis.J Mol Microbiol Biotechnol. 2001 Apr;3(2):219-24. J Mol Microbiol Biotechnol. 2001. PMID: 11321577 Review.
-
Antimicrobial peptide resistance in Neisseria meningitidis.Biochim Biophys Acta. 2015 Nov;1848(11 Pt B):3026-31. doi: 10.1016/j.bbamem.2015.05.006. Epub 2015 May 19. Biochim Biophys Acta. 2015. PMID: 26002321 Free PMC article. Review.
Cited by
-
Modulation of the balance of fatty acid production and secretion is crucial for enhancement of growth and productivity of the engineered mutant of the cyanobacterium Synechococcus elongatus.Biotechnol Biofuels. 2016 Apr 23;9:91. doi: 10.1186/s13068-016-0506-1. eCollection 2016. Biotechnol Biofuels. 2016. PMID: 27110287 Free PMC article.
-
MtrR control of a transcriptional regulatory pathway in Neisseria meningitidis that influences expression of a gene (nadA) encoding a vaccine candidate.PLoS One. 2013;8(2):e56097. doi: 10.1371/journal.pone.0056097. Epub 2013 Feb 8. PLoS One. 2013. PMID: 23409129 Free PMC article.
-
In the NadR regulon, adhesins and diverse meningococcal functions are regulated in response to signals in human saliva.J Bacteriol. 2012 Jan;194(2):460-74. doi: 10.1128/JB.06161-11. Epub 2011 Nov 11. J Bacteriol. 2012. PMID: 22081399 Free PMC article.
-
Deep sequencing-based analysis of the anaerobic stimulon in Neisseria gonorrhoeae.BMC Genomics. 2011 Jan 20;12:51. doi: 10.1186/1471-2164-12-51. BMC Genomics. 2011. PMID: 21251255 Free PMC article.
-
Arachidonic Acid Stress Impacts Pneumococcal Fatty Acid Homeostasis.Front Microbiol. 2018 May 11;9:813. doi: 10.3389/fmicb.2018.00813. eCollection 2018. Front Microbiol. 2018. PMID: 29867785 Free PMC article.
References
-
- Carty, S. M., K. R. Sreekumar, and C. R. Raetz. 1999. Effect of cold shock on lipid A biosynthesis in Escherichia coli. Induction at 12°C of an acyltransferase specific for palmitoleoyl-acyl carrier protein. J. Biol. Chem. 274:9677-9685. - PubMed
-
- Claus, H., R. Borrow, M. Achtman, G. Morelli, C. Kantelberg, E. Longworth, M. Frosch, and U. Vogel. 2004. Genetics of capsule O-acetylation in serogroup C, W-135 and Y meningococci. Mol. Microbiol. 51:227-239. - PubMed
-
- Claus, H., M. C. Maiden, D. J. Wilson, N. D. McCarthy, K. A. Jolley, R. Urwin, F. Hessler, M. Frosch, and U. Vogel. 2005. Genetic analysis of meningococci carried by children and young adults. J. Infect. Dis. 191:1263-1271. - PubMed
-
- Clementz, T., J. J. Bednarski, and C. R. Raetz. 1996. Function of the htrB high temperature requirement gene of Escherichia coli in the acylation of lipid A: HtrB catalyzed incorporation of laurate. J. Biol. Chem. 271:12095-12102. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

