Plk4 is required for cytokinesis and maintenance of chromosomal stability
- PMID: 20348415
- PMCID: PMC2872425
- DOI: 10.1073/pnas.0910941107
Plk4 is required for cytokinesis and maintenance of chromosomal stability
Abstract
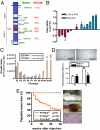
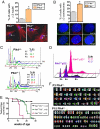
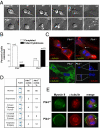
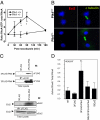
Aneuploidy is a characteristic feature of established cancers and can promote tumor development. Aneuploidy may arise directly, through unequal distribution of chromosomes into daughter cells, or indirectly, through a tetraploid intermediate. The polo family kinase Plk4/Sak is required for late mitotic progression and is haploinsufficient for tumor suppression in mice. Here we show that loss of heterozygosity (LOH) occurs at the Plk4 locus in 50% of human hepatocellular carcinomas (HCC) and is present even in preneoplastic cirrhotic liver nodules. LOH at Plk4 is associated with reduced Plk4 expression in HCC tumors but not with mutations in the remaining allele. Plk4(+/-) murine embryonic fibroblasts (MEFs) at early passage show a high incidence of multinucleation, supernumerary centrosomes, and a near-tetraploid karyotype. Underlying these phenotypes is a high rate of primary cytokinesis failure, associated with aberrant actomyosin ring formation, reduced RhoA activation, and failure to localize the RhoA guanine nucleotide exchange factor Ect2 to the spindle midbody. We further show that Plk4 normally localizes to the midbody and binds to and phosphorylates Ect2 in vitro. With serial passaging Plk4(+/-) MEFs rapidly immortalize, acquiring an increasing burden of nonclonal and clonal gross chromosomal irregularities, and form tumors in vivo. Our results indicate that haploid levels of Plk4 disrupt RhoGTPase function during cytokinesis, resulting in aneuploidy and tumorigenesis, thus implicating early LOH at Plk4 as one of the drivers of human hepatocellular carcinogenesis. These findings represent an advance in our understanding of genetic predisposition to HCC, which continues to increase in incidence globally and particularly in North America.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Polo-like kinase 4 controls centriole duplication but does not directly regulate cytokinesis.Mol Biol Cell. 2012 May;23(10):1838-45. doi: 10.1091/mbc.E11-12-1043. Epub 2012 Mar 28. Mol Biol Cell. 2012. PMID: 22456511 Free PMC article.
-
Plk4 haploinsufficiency causes mitotic infidelity and carcinogenesis.Nat Genet. 2005 Aug;37(8):883-8. doi: 10.1038/ng1605. Epub 2005 Jul 17. Nat Genet. 2005. PMID: 16025114
-
A novel role for Plk4 in regulating cell spreading and motility.Oncogene. 2015 Jun;34(26):3441-51. doi: 10.1038/onc.2014.275. Epub 2014 Sep 1. Oncogene. 2015. PMID: 25174401
-
Sak/Plk4 and mitotic fidelity.Oncogene. 2005 Jan 10;24(2):306-12. doi: 10.1038/sj.onc.1208275. Oncogene. 2005. PMID: 15640847 Review.
-
Mitotic kinases: the key to duplication, segregation, and cytokinesis errors, chromosomal instability, and oncogenesis.Pharmacol Ther. 2006 Sep;111(3):974-84. doi: 10.1016/j.pharmthera.2006.02.006. Epub 2006 Apr 17. Pharmacol Ther. 2006. PMID: 16603252 Review.
Cited by
-
Seize the engine: Emerging cell cycle targets in breast cancer.Clin Transl Med. 2024 Jan;14(1):e1544. doi: 10.1002/ctm2.1544. Clin Transl Med. 2024. PMID: 38264947 Free PMC article. Review.
-
Common variants spanning PLK4 are associated with mitotic-origin aneuploidy in human embryos.Science. 2015 Apr 10;348(6231):235-8. doi: 10.1126/science.aaa3337. Science. 2015. PMID: 25859044 Free PMC article.
-
Polo-like kinase 4 controls centriole duplication but does not directly regulate cytokinesis.Mol Biol Cell. 2012 May;23(10):1838-45. doi: 10.1091/mbc.E11-12-1043. Epub 2012 Mar 28. Mol Biol Cell. 2012. PMID: 22456511 Free PMC article.
-
High PLK4 expression promotes tumor progression and induces epithelial‑mesenchymal transition by regulating the Wnt/β‑catenin signaling pathway in colorectal cancer.Int J Oncol. 2019 Feb;54(2):479-490. doi: 10.3892/ijo.2018.4659. Epub 2018 Dec 10. Int J Oncol. 2019. PMID: 30570110 Free PMC article.
-
Male hypogonadism and germ cell loss caused by a mutation in Polo-like kinase 4.Endocrinology. 2011 Oct;152(10):3975-85. doi: 10.1210/en.2011-1106. Epub 2011 Jul 26. Endocrinology. 2011. PMID: 21791561 Free PMC article.
References
-
- Weaver BA, Silk AD, Montagna C, Verdier-Pinard P, Cleveland DW. Aneuploidy acts both oncogenically and as a tumor suppressor. Cancer Cell. 2007;11:25–36. - PubMed
-
- Fujiwara T, et al. Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature. 2005;437:1043–1047. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

