Induction of TGF-beta 1, not regulatory T cells, impairs antiviral immunity in the lung following bone marrow transplant
- PMID: 20348421
- PMCID: PMC3314499
- DOI: 10.4049/jimmunol.0901871
Induction of TGF-beta 1, not regulatory T cells, impairs antiviral immunity in the lung following bone marrow transplant
Abstract
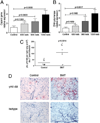
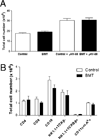
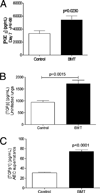
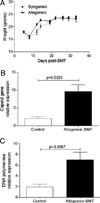
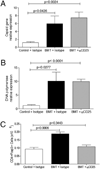
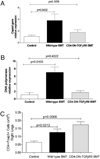
Patients receiving hematopoietic stem cell transplantation or bone marrow transplantation (BMT) as therapy for various malignancies or autoimmune diseases have an increased risk for infectious complications posttransplant, especially in the lung. We have used BMT in mice and murine gammaherpesvirus, gammaHV-68, to study the efficacy of adaptive immune responses post-BMT. Five weeks posttransplant, mice have fully reconstituted their hematopoietic lineages in both the lung and periphery. When challenged with virus, however, BMT mice have a reduced ability to clear lytic virus from the lung. Defective viral control in BMT mice is not related to impaired leukocyte recruitment or defective APC function. Rather, BMT mice are characterized by defective CD4 cell proliferation, skewing of effector CD4 T cells from a Th1 to a Th17 phenotype, and an immunosuppressive lung environment at the time of infection that includes overexpression of TGF-beta1 and PGE(2) and increased numbers of regulatory T cells. Neither indomethacin treatment to block PG synthesis nor anti-CD25 depletion of regulatory T cells improved antiviral host defense post-BMT. Transplanting mice with transgenic bone marrow expressing a dominant-negative TGF-betaRII under the permissive CD4 promoter created mice in which effector CD4 and CD8 cells were unresponsive to TGF-beta1. Mice with TGF-beta1-nonresponsive effector T cells had restored antiviral immunity and improved Th1 responses post-BMT. Thus, our results indicate that overexpression of TGF-beta1 following myeloablative conditioning post-BMT results in impaired effector T cell responses to viral infection.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures






Comment in
-
Comment on "Induction of TGF-beta 1, not regulatory T cells, impairs antiviral immunity in the lung following bone marrow transplant".J Immunol. 2010 Aug 1;185(3):1350-1; author reply 1351. doi: 10.4049/jimmunol.1090060. J Immunol. 2010. PMID: 20660356 No abstract available.
Similar articles
-
Bone marrow transplantation alters lung antigen-presenting cells to promote TH17 response and the development of pneumonitis and fibrosis following gammaherpesvirus infection.Mucosal Immunol. 2016 May;9(3):610-20. doi: 10.1038/mi.2015.85. Epub 2015 Sep 16. Mucosal Immunol. 2016. PMID: 26376362 Free PMC article.
-
Comment on "Induction of TGF-beta 1, not regulatory T cells, impairs antiviral immunity in the lung following bone marrow transplant".J Immunol. 2010 Aug 1;185(3):1350-1; author reply 1351. doi: 10.4049/jimmunol.1090060. J Immunol. 2010. PMID: 20660356 No abstract available.
-
Bone marrow transplant-induced alterations in Notch signaling promote pathologic Th17 responses to γ-herpesvirus infection.Mucosal Immunol. 2018 May;11(3):881-893. doi: 10.1038/mi.2017.85. Epub 2017 Oct 18. Mucosal Immunol. 2018. PMID: 29044226 Free PMC article.
-
Immune control of mammalian gamma-herpesviruses: lessons from murid herpesvirus-4.J Gen Virol. 2009 Oct;90(Pt 10):2317-2330. doi: 10.1099/vir.0.013300-0. Epub 2009 Jul 15. J Gen Virol. 2009. PMID: 19605591 Review.
-
Pleiotropic effects of transforming growth factor-β in hematopoietic stem-cell transplantation.Transplantation. 2010 Dec 15;90(11):1139-44. doi: 10.1097/TP.0b013e3181efd018. Transplantation. 2010. PMID: 20671593 Free PMC article. Review.
Cited by
-
Defective pulmonary innate immune responses post-stem cell transplantation; review and results from one model system.Front Immunol. 2013 May 24;4:126. doi: 10.3389/fimmu.2013.00126. eCollection 2013. Front Immunol. 2013. PMID: 23745124 Free PMC article.
-
Acquired and hereditary bone marrow failure: A mitochondrial perspective.Front Oncol. 2022 Nov 2;12:1048746. doi: 10.3389/fonc.2022.1048746. eCollection 2022. Front Oncol. 2022. PMID: 36408191 Free PMC article. Review.
-
Fates of CD4+ T cells in a tolerant environment depend on timing and place of antigen exposure.Am J Transplant. 2012 Mar;12(3):576-89. doi: 10.1111/j.1600-6143.2011.03879.x. Epub 2011 Dec 17. Am J Transplant. 2012. PMID: 22176785 Free PMC article.
-
The Histone Methyltransferase Setdb2 Modulates Macrophage Phenotype and Uric Acid Production in Diabetic Wound Repair.Immunity. 2019 Aug 20;51(2):258-271.e5. doi: 10.1016/j.immuni.2019.06.015. Epub 2019 Jul 23. Immunity. 2019. PMID: 31350176 Free PMC article.
-
Bone marrow transplantation alters lung antigen-presenting cells to promote TH17 response and the development of pneumonitis and fibrosis following gammaherpesvirus infection.Mucosal Immunol. 2016 May;9(3):610-20. doi: 10.1038/mi.2015.85. Epub 2015 Sep 16. Mucosal Immunol. 2016. PMID: 26376362 Free PMC article.
References
-
- Horowitz MM, Loberiza FR, Bredeson CN, Rizzo JD, Nugent ML. Transplant registries: guiding clinical decisions and improving outcomes. Oncology (Williston Park) 2001;15:649–659. discussion 663–644, 666. - PubMed
-
- Copelan EA. Hematopoietic stem-cell transplantation. N. Engl. J. Med. 2006;354:1813–1826. - PubMed
-
- Soubani AO, Miller KB, Hassoun PM. Pulmonary complications of bone marrow transplantation. Chest. 1996;109:1066–1077. - PubMed
-
- Wald A, Leisenring W, van Burik JA, Bowden RA. Epidemiology of Aspergillus infections in a large cohort of patients undergoing bone marrow transplantation. J. Infect. Dis. 1997;175:1459–1466. - PubMed
-
- Lossos IS, Breuer R, Or R, Strauss N, Elishoov H, Naparstek E, Aker M, Nagler A, Moses AE, Shapiro M, et al. Bacterial pneumonia in recipients of bone marrow transplantation: a five-year prospective study. Transplantation. 1995;60:672–678. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

