IL-6 increases B-cell IgG production in a feed-forward proinflammatory mechanism to skew hematopoiesis and elevate myeloid production
- PMID: 20351305
- PMCID: PMC3790945
- DOI: 10.1182/blood-2009-07-230631
IL-6 increases B-cell IgG production in a feed-forward proinflammatory mechanism to skew hematopoiesis and elevate myeloid production
Abstract
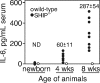
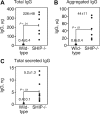
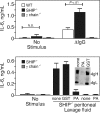
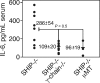
Src homology 2 domain-containing inositol 5-phosphatase (SHIP(-/-)) animals display an age-related increase in interleukin-6 (IL-6), a decrease in B lymphopoiesis, and an elevation in myelopoiesis. We investigated the origin of the IL-6 production and show that it is largely produced by peritoneal and splenic macrophages. IL-6 production by these macrophages is not a direct result of the loss of SHIP: IL-6 production is not spontaneous, is absent from bone marrow-derived macrophages, declines with prolonged culture of macrophages, and requires a stimulus present in vivo. The IL-6-rich peritoneal cavity of SHIP(-/-) mice shows more than 700-fold more immunoglobulin G (IgG) than wild-type, approximately 20% of which is aggregated or in an immune complex and contains B220(+) cells that secrete IgG. The SHIP-deficient peritoneal macrophages show evidence of IgG receptor stimulation. Animals lacking both the signal-transducing gamma-chain of IgG receptors and SHIP or Ig and SHIP produce less IL-6. The data indicate a feed-forward process in which peripheral macrophages, responding through IgG receptors to secreted IgG, produce IL-6, to support further B-cell production of IgG. Because of the proinflammatory phenotype of SHIP(-/-) animals, these findings emphasize the importance of IL-6-neutralizing strategies in autoimmune and proinflammatory diseases.
Figures


Similar articles
-
SHIP-1 deficiency in the myeloid compartment is insufficient to induce myeloid expansion or chronic inflammation.Genes Immun. 2014 Apr;15(4):233-40. doi: 10.1038/gene.2014.9. Epub 2014 Mar 6. Genes Immun. 2014. PMID: 24598798
-
Differential roles for the inositol phosphatase SHIP in the regulation of macrophages and lymphocytes.Immunol Res. 2009;43(1-3):243-51. doi: 10.1007/s12026-008-8078-1. Immunol Res. 2009. PMID: 18989630 Free PMC article. Review.
-
Src homology 2-containing 5-inositol phosphatase (SHIP) suppresses an early stage of lymphoid cell development through elevated interleukin-6 production by myeloid cells in bone marrow.J Exp Med. 2004 Jan 19;199(2):243-54. doi: 10.1084/jem.20031193. Epub 2004 Jan 12. J Exp Med. 2004. PMID: 14718513 Free PMC article.
-
The inositol phosphatase SHIP-2 down-regulates FcgammaR-mediated phagocytosis in murine macrophages independently of SHIP-1.Blood. 2006 Jan 15;107(2):813-20. doi: 10.1182/blood-2005-05-1841. Epub 2005 Sep 22. Blood. 2006. PMID: 16179375 Free PMC article.
-
Role of Src homology 2-containing-inositol 5'-phosphatase (SHIP) in mast cells and macrophages.Biochem Soc Trans. 2003 Feb;31(Pt 1):286-91. doi: 10.1042/bst0310286. Biochem Soc Trans. 2003. PMID: 12546703 Review.
Cited by
-
The Onset of Antinuclear Antibodies (ANAs) as a Potential Risk Factor for Mortality and Morbidity in COVID-19 Patients: A Single-Center Retrospective Study.Biomedicines. 2024 Jun 13;12(6):1306. doi: 10.3390/biomedicines12061306. Biomedicines. 2024. PMID: 38927513 Free PMC article.
-
B cell checkpoints in autoimmune rheumatic diseases.Nat Rev Rheumatol. 2019 May;15(5):303-315. doi: 10.1038/s41584-019-0211-0. Nat Rev Rheumatol. 2019. PMID: 30967621 Review.
-
Biomolecular basis of the role of diabetes mellitus in osteoporosis and bone fractures.World J Diabetes. 2013 Aug 15;4(4):101-13. doi: 10.4239/wjd.v4.i4.101. World J Diabetes. 2013. PMID: 23961320 Free PMC article.
-
Cell Intrinsic IL-38 Affects B Cell Differentiation and Antibody Production.Int J Mol Sci. 2023 Mar 16;24(6):5676. doi: 10.3390/ijms24065676. Int J Mol Sci. 2023. PMID: 36982750 Free PMC article.
-
Inhibitor and activator: dual functions for SHIP in immunity and cancer.Ann N Y Acad Sci. 2011 Jan;1217:1-17. doi: 10.1111/j.1749-6632.2010.05869.x. Epub 2010 Dec 13. Ann N Y Acad Sci. 2011. PMID: 21155837 Free PMC article. Review.
References
-
- Kishimoto T. Interleukin-6: from basic science to medicine—40 years in immunology. Annu Rev Immunol. 2005;23:1–21. - PubMed
-
- Yoshizaki K, Nakagawa T, Fukunaga K, Tseng LT, Yamamura Y, Kishimoto T. Isolation and characterization of B cell differentiation factor (BCDF) secreted from a human B lymphoblastoid cell line. J Immunol. 1984;132(6):2948–2954. - PubMed
-
- Chun HY, Chung JW, Kim HA, et al. Cytokine IL-6 and IL-10 as biomarkers in systemic lupus erythematosus. J Clin Immunol. 2007;27(5):461–466. - PubMed
-
- Knudsen LS, Klarlund M, Skjodt H, et al. Biomarkers of inflammation in patients with unclassified polyarthritis and early rheumatoid arthritis: relationship to disease activity and radiographic outcome. J Rheumatol. 2008;35(7):1277–1287. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

