Pilot study of a (213)bismuth-labeled anti-CD45 mAb as a novel nonmyeloablative conditioning for DLA-haploidentical littermate hematopoietic transplantation
- PMID: 20351629
- PMCID: PMC2932447
- DOI: 10.1097/TP.0b013e3181d98c3d
Pilot study of a (213)bismuth-labeled anti-CD45 mAb as a novel nonmyeloablative conditioning for DLA-haploidentical littermate hematopoietic transplantation
Abstract
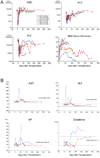
Background: A pilot study was conducted to determine whether conditioning using selective targeting of hematopoietic cells with an alpha-particle emitter, bismuth-213 ((213)Bi)-labeled anti-CD45 monoclonal antibody (mAb) is sufficient to overcome the major histocompatibility barrier in a canine model of dog leukocyte antigen-haploidentical hematopoietic cell transplantation (HCT).
Methods: Six dogs were administered 0.5 mg/kg (213)Bi-labeled anti-CD45 mAb (dose (213)Bi=2.26-4.9 mCi/kg) in six to eight injections. For postgrafting immunosuppression, all dogs received cyclosporine and mycophenolate mofetil.
Results: All dogs had initial donor engraftment, with three of six dogs having sustained engraftment to last point of follow-up. Two dogs receiving 2.26 and 3.25 mCi/kg of (213)Bi rejected their grafts at day +127 and +125, respectively, whereas dogs receiving (213)Bi doses of 3.3 mCi/kg or greater achieved high level donor chimerism.
Conclusion: The results suggest that nonmyeloablative conditioning with (213)Bi-labeled anti-CD45 mAb could be applicable to major histocompatibility haploidentical HCT without excessive nonhematologic regimen-related toxicity.
Figures
Similar articles
-
Radioimmunotherapy with bismuth-213 as conditioning for nonmyeloablative allogeneic hematopoietic cell transplantation in dogs: a dose deescalation study.Transplantation. 2004 Aug 15;78(3):352-9. doi: 10.1097/01.tp.0000128853.62545.b2. Transplantation. 2004. PMID: 15316362
-
Bismuth 213-labeled anti-CD45 radioimmunoconjugate to condition dogs for nonmyeloablative allogeneic marrow grafts.Blood. 2002 Jul 1;100(1):318-26. doi: 10.1182/blood-2001-12-0322. Blood. 2002. PMID: 12070043
-
Dog leukocyte antigen-haploidentical stem cell allografts after anti-CD44 therapy and nonmyeloablative conditioning in a preclinical canine model.Transplantation. 2006 Aug 15;82(3):332-9. doi: 10.1097/01.tp.0000228908.10775.b0. Transplantation. 2006. PMID: 16906030
-
Alloreactivity as therapeutic principle in the treatment of hematologic malignancies. Studies of clinical and immunologic aspects of allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning.Dan Med Bull. 2007 May;54(2):112-39. Dan Med Bull. 2007. PMID: 17521527 Review.
-
Radioimmunotherapy as non-myeloablative conditioning for allogeneic marrow transplantation.Leuk Lymphoma. 2006 Jul;47(7):1205-14. doi: 10.1080/00423110500485822. Leuk Lymphoma. 2006. PMID: 16923548 Review.
Cited by
-
Animal Models for Preclinical Development of Allogeneic Hematopoietic Cell Transplantation.ILAR J. 2018 Dec 31;59(3):263-275. doi: 10.1093/ilar/ily006. ILAR J. 2018. PMID: 30010833 Free PMC article.
-
Durable donor engraftment after radioimmunotherapy using α-emitter astatine-211-labeled anti-CD45 antibody for conditioning in allogeneic hematopoietic cell transplantation.Blood. 2012 Feb 2;119(5):1130-8. doi: 10.1182/blood-2011-09-380436. Epub 2011 Dec 1. Blood. 2012. PMID: 22134165 Free PMC article.
-
211At-Labeled Anti-CD45 Antibody as a Nonmyeloablative Conditioning for Canine DLA-Haploidentical Stem Cell Transplantation.J Nucl Med. 2024 Sep 3;65(9):1443-1449. doi: 10.2967/jnumed.124.267540. J Nucl Med. 2024. PMID: 39025648 Free PMC article.
-
Conditioning with α-emitter based radioimmunotherapy in canine allogeneic hematopoietic cell transplantation.Chimerism. 2012 Apr-Jun;3(2):40-2. doi: 10.4161/chim.20726. Epub 2012 Apr 1. Chimerism. 2012. PMID: 22772070 Free PMC article.
-
The road to purified hematopoietic stem cell transplants is paved with antibodies.Curr Opin Immunol. 2012 Oct;24(5):640-8. doi: 10.1016/j.coi.2012.08.002. Epub 2012 Aug 29. Curr Opin Immunol. 2012. PMID: 22939368 Free PMC article. Review.
References
-
- Matthews DC, Appelbaum FR, Eary JF, et al. Development of a marrow transplant regimen for acute leukemia using targeted hematopoietic irradiation delivered by 131I-labeled anti-CD45 antibody, combined with cyclophosphamide and total body irradiation. Blood. 1995;85:1122–1131. - PubMed
-
- Matthews DC, Martin PJ, Nourigat C, Appelbaum FR, Fisher DR, Bernstein ID. Marrow ablative and immunosuppressive effects of 131 I-anti-CD45 antibody in congenic and H2-mismatched murine transplant models. Blood. 1999;93:737–745. - PubMed
-
- Matthews DC, Appelbaum FR, Eary JF, et al. Phase I study of 131I-Anti-CD45 antibody plus cyclophosphamide and total body irradiation for advanced acute leukemia and myelodysplastic syndrome. Blood. 1999;94:1237–1247. - PubMed
-
- Sandmaier BM, Bethge WA, Wilbur DS, et al. Bismuth 213-labeled anti-CD45 radioimmunoconjugate to condition dogs for nonmyeloablative allogeneic marrow grafts. Blood. 2002;100:318–326. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

