The MinCDJ system in Bacillus subtilis prevents minicell formation by promoting divisome disassembly
- PMID: 20352045
- PMCID: PMC2844427
- DOI: 10.1371/journal.pone.0009850
The MinCDJ system in Bacillus subtilis prevents minicell formation by promoting divisome disassembly
Abstract
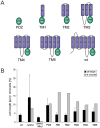
Background: Cell division in Bacillus subtilis takes place precisely at midcell, through the action of Noc, which prevents division from occurring over the nucleoids, and the Min system, which prevents cell division from taking place at the poles. Originally it was thought that the Min system acts directly on FtsZ, preventing the formation of a Z-ring and, therefore, the formation of a complete cytokinetic ring at the poles. Recently, a new component of the B. subtilis Min system was identified, MinJ, which acts as a bridge between DivIVA and MinCD.
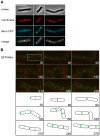
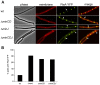
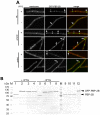
Methodology/principal findings: We used fluorescence microscopy and molecular genetics to examine the molecular role of MinJ. We found that in the absence of a functional Min system, FtsA, FtsL and PBP-2B remain associated with completed division sites. Evidence is provided that MinCDJ are responsible for the failure of these proteins to localize properly, indicating that MinCDJ can act on membrane integral components of the divisome.
Conclusions/significance: Taken together, we postulate that the main function of the Min system is to prevent minicell formation adjacent to recently completed division sites by promoting the disassembly of the cytokinetic ring, thereby ensuring that cell division occurs only once per cell cycle. Thus, the role of the Min system in rod-shaped bacteria seems not to be restricted to an inhibitory function on FtsZ polymerization, but can act on different levels of the divisome.
Conflict of interest statement
Figures



Similar articles
-
Cellular architecture mediates DivIVA ultrastructure and regulates min activity in Bacillus subtilis.mBio. 2011 Nov 22;2(6):e00257-11. doi: 10.1128/mBio.00257-11. Print 2011. mBio. 2011. PMID: 22108385 Free PMC article.
-
A novel component of the division-site selection system of Bacillus subtilis and a new mode of action for the division inhibitor MinCD.Mol Microbiol. 2008 Dec;70(6):1556-69. doi: 10.1111/j.1365-2958.2008.06501.x. Epub 2008 Oct 23. Mol Microbiol. 2008. PMID: 19019154
-
Bacillus subtilis MinC destabilizes FtsZ-rings at new cell poles and contributes to the timing of cell division.Genes Dev. 2008 Dec 15;22(24):3475-88. doi: 10.1101/gad.1732408. Genes Dev. 2008. PMID: 19141479 Free PMC article.
-
Cell Cycle Machinery in Bacillus subtilis.Subcell Biochem. 2017;84:67-101. doi: 10.1007/978-3-319-53047-5_3. Subcell Biochem. 2017. PMID: 28500523 Free PMC article. Review.
-
Bacterial cell division: the mechanism and its precison.Int Rev Cytol. 2006;253:27-94. doi: 10.1016/S0074-7696(06)53002-5. Int Rev Cytol. 2006. PMID: 17098054 Review.
Cited by
-
SecA is required for membrane targeting of the cell division protein DivIVA in vivo.Front Microbiol. 2014 Feb 14;5:58. doi: 10.3389/fmicb.2014.00058. eCollection 2014. Front Microbiol. 2014. PMID: 24592260 Free PMC article.
-
Imaging DivIVA dynamics using photo-convertible and activatable fluorophores in Bacillus subtilis.Front Microbiol. 2014 Feb 18;5:59. doi: 10.3389/fmicb.2014.00059. eCollection 2014. Front Microbiol. 2014. PMID: 24600441 Free PMC article.
-
Dynamics of the Bacillus subtilis Min System.mBio. 2021 Apr 13;12(2):e00296-21. doi: 10.1128/mBio.00296-21. mBio. 2021. PMID: 33849976 Free PMC article.
-
The Division Defect of a Bacillus subtilis minD noc Double Mutant Can Be Suppressed by Spx-Dependent and Spx-Independent Mechanisms.J Bacteriol. 2021 Aug 20;203(18):e0024921. doi: 10.1128/JB.00249-21. Epub 2021 Aug 20. J Bacteriol. 2021. PMID: 34181483 Free PMC article.
-
Metabolic Perturbations in a Bacillus subtilis clpP Mutant during Glucose Starvation.Metabolites. 2017 Nov 24;7(4):63. doi: 10.3390/metabo7040063. Metabolites. 2017. PMID: 29186773 Free PMC article.
References
-
- Harry E, Monahan L, Thompson L. Bacterial cell division: the mechanism and its precison. Int Rev Cytol. 2006;253:27–94. - PubMed
-
- Goehring NW, Beckwith J. Diverse paths to midcell: assembly of the bacterial cell division machinery. Curr Biol. 2005;15:R514–526. - PubMed
-
- Adams DW, Errington J. Bacterial cell division: assembly, maintenance and disassembly of the Z ring. Nat Rev Microbiol. 2009;7:642–653. - PubMed
-
- Bi EF, Lutkenhaus J. FtsZ ring structure associated with division in Escherichia coli. Nature. 1991;354:161–164. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

