In the absence of frazzled over-expression of Abelson tyrosine kinase disrupts commissure formation and causes axons to leave the embryonic CNS
- PMID: 20352105
- PMCID: PMC2843715
- DOI: 10.1371/journal.pone.0009822
In the absence of frazzled over-expression of Abelson tyrosine kinase disrupts commissure formation and causes axons to leave the embryonic CNS
Abstract
Background: In the Drosophila embryonic nerve cord, the formation of commissures require both the chemoattractive Netrin receptor Frazzled (Fra) and the Abelson (Abl) cytoplasmic tyrosine kinase. Abl binds to the cytoplasmic domain of Fra and loss-of-function mutations in abl enhance fra-dependent commissural defects. To further test Abl's role in attractive signaling, we over-expressed Abl in Fra mutants anticipating rescue of commissures.
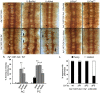
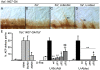
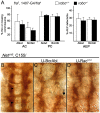
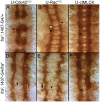
Methodology/principal findings: The Gal4-UAS system was used to pan-neurally over-express Abl in homozygous fra embryos. Surprisingly, this led to a significant decrease in both posterior and anterior commissure formation and induced some commissural and longitudinal axons to project beyond the CNS/PNS border. Re-expressing wild-type Fra, or Fra mutants with a P-motif deleted, revert both commissural and exiting phenotypes, indicating that Fra is required but not a specific P-motif. This is supported by S2 cell experiments demonstrating that Abl binds to Fra independent of any specific P-motif and that Fra continues to be phosphorylated when individual P-motifs are removed. Decreasing midline repulsion by reducing Robo signaling had no effect on the Abl phenotype and the phenotypes still occur in a Netrin mutant. Pan-neural over-expression of activated Rac or Cdc42 in a fra mutant also induced a significant loss in commissures, but axons did not exit the CNS.
Conclusion/significance: Taken together, these data suggest that Fra activity is required to correctly regulate Abl-dependent cytoskeletal dynamics underlying commissure formation. In the absence of Fra, increased Abl activity appears to be incorrectly utilized downstream of other guidance receptors resulting in a loss of commissures and the abnormal projections of some axons beyond the CNS/PNS border.
Conflict of interest statement
Figures



Similar articles
-
The Abelson tyrosine kinase, the Trio GEF and Enabled interact with the Netrin receptor Frazzled in Drosophila.Development. 2005 Apr;132(8):1983-94. doi: 10.1242/dev.01736. Development. 2005. PMID: 15790972
-
Frazzled cytoplasmic P-motifs are differentially required for axon pathway formation in the Drosophila embryonic CNS.Int J Dev Neurosci. 2008 Nov;26(7):753-61. doi: 10.1016/j.ijdevneu.2008.07.004. Epub 2008 Jul 11. Int J Dev Neurosci. 2008. PMID: 18674607
-
Frazzled regulation of myosin II activity in the Drosophila embryonic CNS.Dev Biol. 2007 Aug 1;308(1):120-32. doi: 10.1016/j.ydbio.2007.05.012. Epub 2007 May 18. Dev Biol. 2007. PMID: 17568577
-
Axon guidance at the midline choice point.Dev Dyn. 2001 Jun;221(2):154-81. doi: 10.1002/dvdy.1143. Dev Dyn. 2001. PMID: 11376484 Review.
-
Embryonic axon guidance: insights from Drosophila and other insects.Curr Opin Insect Sci. 2016 Dec;18:11-16. doi: 10.1016/j.cois.2016.08.007. Epub 2016 Aug 24. Curr Opin Insect Sci. 2016. PMID: 27939705 Review.
Cited by
-
Midline axon guidance in the Drosophila embryonic central nervous system.Semin Cell Dev Biol. 2019 Jan;85:13-25. doi: 10.1016/j.semcdb.2017.11.029. Epub 2017 Nov 27. Semin Cell Dev Biol. 2019. PMID: 29174915 Free PMC article. Review.
-
Control of dendritic morphogenesis by Trio in Drosophila melanogaster.PLoS One. 2012;7(3):e33737. doi: 10.1371/journal.pone.0033737. Epub 2012 Mar 15. PLoS One. 2012. PMID: 22438988 Free PMC article.
-
The first quarter of the C-terminal domain of Abelson regulates the WAVE regulatory complex and Enabled in axon guidance.Neural Dev. 2020 May 2;15(1):7. doi: 10.1186/s13064-020-00144-8. Neural Dev. 2020. PMID: 32359359 Free PMC article.
-
Long disordered regions of the C-terminal domain of Abelson tyrosine kinase have specific and additive functions in regulation and axon localization.PLoS One. 2017 Dec 12;12(12):e0189338. doi: 10.1371/journal.pone.0189338. eCollection 2017. PLoS One. 2017. PMID: 29232713 Free PMC article.
-
The Role of Apoptotic Signaling in Axon Guidance.J Dev Biol. 2018 Oct 18;6(4):24. doi: 10.3390/jdb6040024. J Dev Biol. 2018. PMID: 30340315 Free PMC article. Review.
References
-
- Seeger M, Tear G, Ferres-Marco D, Goodman CS. Mutations affecting growth cone guidance in Drosophila: genes necessary for guidance toward or away from the midline. Neuron. 1993;10:409–426. - PubMed
-
- Garbe DS, Bashaw GJ. Axon guidance at the midline: from mutants to mechanisms. Crit Rev Biochem Mol Biol. 2004;39:319–341. - PubMed
-
- Furrer MP, Kim S, Wolf B, Chiba A. Robo and Frazzled/DCC mediate dendritic guidance at the CNS midline. Nat Neurosci. 2003;6:223–230. - PubMed
-
- Furrer MP, Vasenkova I, Kamiyama D, Rosado Y, Chiba A. Slit and Robo control the development of dendrites in Drosophila CNS. Development. 2007;134:3795–3804. - PubMed
-
- Harris R, Sabatelli LM, Seeger MA. Guidance cues at the Drosophila CNS midline: identification and characterization of two Drosophila Netrin/UNC-6 homologs. Neuron. 1996;17:217–228. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

