Probing native protein structures by chemical cross-linking, mass spectrometry, and bioinformatics
- PMID: 20360032
- PMCID: PMC2938055
- DOI: 10.1074/mcp.R000001-MCP201
Probing native protein structures by chemical cross-linking, mass spectrometry, and bioinformatics
Abstract
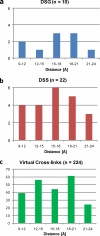
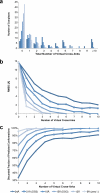
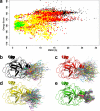
Chemical cross-linking of reactive groups in native proteins and protein complexes in combination with the identification of cross-linked sites by mass spectrometry has been in use for more than a decade. Recent advances in instrumentation, cross-linking protocols, and analysis software have led to a renewed interest in this technique, which promises to provide important information about native protein structure and the topology of protein complexes. In this article, we discuss the critical steps of chemical cross-linking and its implications for (structural) biology: reagent design and cross-linking protocols, separation and mass spectrometric analysis of cross-linked samples, dedicated software for data analysis, and the use of cross-linking data for computational modeling. Finally, the impact of protein cross-linking on various biological disciplines is highlighted.
Figures

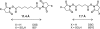

References
-
- Young M. M., Tang N., Hempel J. C., Oshiro C. M., Taylor E. W., Kuntz I. D., Gibson B. W., Dollinger G. (2000) High throughput protein fold identification by using experimental constraints derived from intramolecular cross-links and mass spectrometry. Proc. Natl. Acad. Sci. U.S.A. 97, 5802–5806 - PMC - PubMed
-
- Back J. W., de Jong L., Muijsers A. O., de Koster C. G. (2003) Chemical cross-linking and mass spectrometry for protein structural modeling. J. Mol. Biol. 331, 303–313 - PubMed
-
- Sinz A. (2006) Chemical cross-linking and mass spectrometry to map three-dimensional protein structures and protein-protein interactions. Mass Spectrom. Rev. 25, 663–682 - PubMed
-
- Lee Y. J. (2008) Mass spectrometric analysis of cross-linking sites for the structure of proteins and protein complexes. Mol. Biosyst. 4, 816–823 - PubMed
-
- Schilling B., Row R. H., Gibson B. W., Guo X., Young M. M. (2003) MS2Assign, automated assignment and nomenclature of tandem mass spectra of chemically crosslinked peptides. J. Am. Soc. Mass Spectrom. 14, 834–850 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

