EphA2 mutation in lung squamous cell carcinoma promotes increased cell survival, cell invasion, focal adhesions, and mammalian target of rapamycin activation
- PMID: 20360610
- PMCID: PMC2881783
- DOI: 10.1074/jbc.M109.075085
EphA2 mutation in lung squamous cell carcinoma promotes increased cell survival, cell invasion, focal adhesions, and mammalian target of rapamycin activation
Abstract
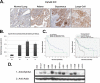
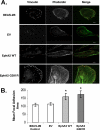
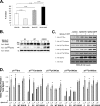
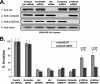
Non-small cell lung cancer (NSCLC) has a poor prognosis and improved therapies are needed. Expression of EphA2 is increased in NSCLC metastases. In this study, we investigated EphA2 mutations in NSCLC and examined molecular pathways involved in NSCLC. Tumor and cell line DNA was sequenced. One EphA2 mutation was modeled by expression in BEAS2B cells, and functional and biochemical studies were conducted. A G391R mutation was detected in H2170 and 2/28 squamous cell carcinoma patient samples. EphA2 G391R caused constitutive activation of EphA2 with increased phosphorylation of Src, cortactin, and p130(Cas). Wild-type (WT) and G391R cells had 20 and 40% increased invasiveness; this was attenuated with knockdown of Src, cortactin, or p130(Cas). WT and G391R cells demonstrated a 70% increase in focal adhesion area. Mammalian target of rapamycin (mTOR) phosphorylation was increased in G391R cells with increased survival (55%) compared with WT (30%) and had increased sensitivity to rapamycin. A recurrent EphA2 mutation is present in lung squamous cell carcinoma and increases tumor invasion and survival through activation of focal adhesions and actin cytoskeletal regulatory proteins as well as mTOR. Further study of EphA2 as a therapeutic target is warranted.
Figures



References
-
- Jemal A., Siegel R., Ward E., Murray T., Xu J., Thun M. J. (2007) CA Cancer J. Clin. 57, 43–66 - PubMed
-
- Naruke T., Goya T., Tsuchiya R., Suemasu K. (1988) J. Thorac. Cardiovasc. Surg. 96, 440–447 - PubMed
-
- Nesbitt J. C., Putnam J. B., Jr., Walsh G. L., Roth J. A., Mountain C. F. (1995) Ann. Thorac. Surg. 60, 466–472 - PubMed
-
- van Rens M. T., de la Rivière A. B., Elbers H. R., van Den Bosch J. M. (2000) Chest. 117, 374–379 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

