Population pharmacokinetics and pharmacodynamic considerations of amodiaquine and desethylamodiaquine in Kenyan adults with uncomplicated malaria receiving artesunate-amodiaquine combination therapy
- PMID: 20368402
- PMCID: PMC2876388
- DOI: 10.1128/AAC.01496-09
Population pharmacokinetics and pharmacodynamic considerations of amodiaquine and desethylamodiaquine in Kenyan adults with uncomplicated malaria receiving artesunate-amodiaquine combination therapy
Abstract
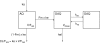
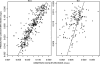
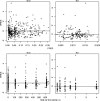
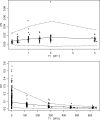
Amodiaquine (AQ) is an antimalarial drug that was frequently combined with artesunate (AS) for the treatment of uncomplicated malaria due to Plasmodium falciparum and is now available as a fixed-dose combination. Despite its widespread use, the simultaneous pharmacokinetics in patients of AQ and its active metabolite, desethylamodiaquine (DAQ), were not characterized to date. The pharmacokinetics of AQ and DAQ in 54 adult patients receiving the AS/AQ combination were therefore investigated by the use of a population approach. AQ followed a 1-compartment model with first-order absorption and elimination, as well as a first-order and irreversible transformation into DAQ, which in turn followed a 2-compartment model with first-order elimination from its central compartment. The mean AQ apparent clearance and distribution volume were 3,410 liters/h and 39,200 liters, respectively. The mean terminal elimination half-life of DAQ was 211 h. Body weight was found to explain the interindividual variability of the apparent volume of distribution of AQ and the elimination rate constant of DAQ. A new dosage form consisting of a fixed-dose combination of AS and AQ was found to have no effect on the pharmacokinetic parameters of AQ and DAQ. All patients achieved parasite clearance within 4 days following the initiation of the treatment, which prevented investigation of the possible relationship between DAQ exposure and treatment outcome. This study provided the first simultaneous pharmacokinetic model for AQ and DAQ.
Figures

Similar articles
-
Fixed dose artesunate amodiaquine - a phase IIb, randomized comparative trial with non-fixed artesunate amodiaquine.Malar J. 2014 Dec 16;13:498. doi: 10.1186/1475-2875-13-498. Malar J. 2014. PMID: 25515698 Free PMC article. Clinical Trial.
-
Population pharmacokinetics of artesunate and amodiaquine in African children.Malar J. 2009 Aug 20;8:200. doi: 10.1186/1475-2875-8-200. Malar J. 2009. PMID: 19691851 Free PMC article. Clinical Trial.
-
Validation and pharmacokinetic application of a high-performance liquid chromatographic technique for determining the concentrations of amodiaquine and its metabolite in plasma of patients treated with oral fixed-dose amodiaquine-artesunate combination in areas of malaria endemicity.Antimicrob Agents Chemother. 2015 Sep;59(9):5114-22. doi: 10.1128/AAC.04957-14. Epub 2015 Apr 20. Antimicrob Agents Chemother. 2015. PMID: 25896711 Free PMC article. Clinical Trial.
-
Therapeutic efficacy of artesunate-amodiaquine combinations and the plasma and saliva concentrations of desethylamodiaquine in children with acute uncomplicated Plasmodium falciparum malaria.Am J Ther. 2013 Jan;20(1):48-56. doi: 10.1097/MJT.0b013e3181ff7c31. Am J Ther. 2013. PMID: 21192244 Clinical Trial.
-
Artesunate-amodiaquine for the treatment of uncomplicated malaria.Expert Opin Investig Drugs. 2007 Jul;16(7):1079-85. doi: 10.1517/13543784.16.7.1079. Expert Opin Investig Drugs. 2007. PMID: 17594191 Review.
Cited by
-
The efficacy and tolerability of artemisinin-piperaquine (Artequick®) versus artesunate-amodiaquine (Coarsucam™) for the treatment of uncomplicated Plasmodium falciparum malaria in south-central Vietnam.Malar J. 2012 Jun 28;11:217. doi: 10.1186/1475-2875-11-217. Malar J. 2012. PMID: 22741618 Free PMC article. Clinical Trial.
-
Population Pharmacokinetic Properties of Sulfadoxine and Pyrimethamine: a Pooled Analysis To Inform Optimal Dosing in African Children with Uncomplicated Malaria.Antimicrob Agents Chemother. 2018 Apr 26;62(5):e01370-17. doi: 10.1128/AAC.01370-17. Print 2018 May. Antimicrob Agents Chemother. 2018. PMID: 29463542 Free PMC article.
-
A randomized trial of artesunate-amodiaquine versus artemether-lumefantrine in Ghanaian paediatric sickle cell and non-sickle cell disease patients with acute uncomplicated malaria.Malar J. 2014 Sep 19;13:369. doi: 10.1186/1475-2875-13-369. Malar J. 2014. PMID: 25236838 Free PMC article. Clinical Trial.
-
Pharmacokinetics of amodiaquine and desethylamodiaquine in pregnant and postpartum women with Plasmodium vivax malaria.Antimicrob Agents Chemother. 2011 Sep;55(9):4338-42. doi: 10.1128/AAC.00154-11. Epub 2011 Jun 27. Antimicrob Agents Chemother. 2011. PMID: 21709098 Free PMC article. Clinical Trial.
-
The cardiovascular effects of amodiaquine and structurally related antimalarials: An individual patient data meta-analysis.PLoS Med. 2021 Sep 7;18(9):e1003766. doi: 10.1371/journal.pmed.1003766. eCollection 2021 Sep. PLoS Med. 2021. PMID: 34492005 Free PMC article.
References
-
- Adjei, G. O., K. Kristensen, B. Q. Goka, L. C. Hoegberg, M. Alifrangis, O. P. Rodrigues, and J. A. Kurtzhals. 2008. Effect of concomitant artesunate administration and cytochrome P4502C8 polymorphisms on the pharmacokinetics of amodiaquine in Ghanaian children with uncomplicated malaria. Antimicrob. Agents Chemother. 52:4400-4406. - PMC - PubMed
-
- Adjei, G. O., J. A. Kurtzhals, O. P. Rodrigues, M. Alifrangis, L. C. Hoegberg, E. D. Kitcher, E. V. Badoe, R. Lamptey, and B. Q. Goka. 2008. Amodiaquine-artesunate vs artemether-lumefantrine for uncomplicated malaria in Ghanaian children: a randomized efficacy and safety trial with one year follow-up. Malar. J. 7:127. - PMC - PubMed
-
- Ashley, E. A., K. Stepniewska, N. Lindegardh, R. McGready, R. Hutagalung, R. Hae, P. Singhasivanon, N. J. White, and F. Nosten. 2006. Population pharmacokinetic assessment of a new regimen of mefloquine used in combination treatment of uncomplicated falciparum malaria. Antimicrob. Agents Chemother. 50:2281-2285. - PMC - PubMed
-
- Aubouy, A., M. Bakary, A. Keundjian, B. Mbomat, J. R. Makita, F. Migot-Nabias, M. Cot, J. Le Bras, and P. Deloron. 2003. Combination of drug level measurement and parasite genotyping data for improved assessment of amodiaquine and sulfadoxine-pyrimethamine efficacies in treating Plasmodium falciparum malaria in Gabonese children. Antimicrob. Agents Chemother. 47:231-237. - PMC - PubMed
-
- Beal, S. L., and L. B. Sheiner. 1991. NONMEM user's guide. NONMEM Project Group, University of California at San Francisco, San Francisco, CA.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

