Phase II clinical trial of sorafenib in metastatic medullary thyroid cancer
- PMID: 20368568
- PMCID: PMC2881718
- DOI: 10.1200/JCO.2009.25.0068
Phase II clinical trial of sorafenib in metastatic medullary thyroid cancer
Abstract
Purpose: Mutations in the RET proto-oncogene and vascular endothelial growth factor receptor (VEGFR) activity are critical in the pathogenesis of medullary thyroid cancer (MTC). Sorafenib, a multikinase inhibitor targeting Ret and VEGFR, showed antitumor activity in preclinical studies of MTC.
Patients and methods: In this phase II trial of sorafenib in patients with advanced MTC, the primary end point was objective response. Secondary end points included toxicity assessment and response correlation with tumor markers, functional imaging, and RET mutations. Using a two-stage design, 16 or 25 patients were to be enrolled onto arms A (hereditary) and B (sporadic). Patients received sorafenib 400 mg orally twice daily.
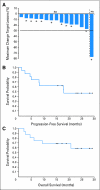
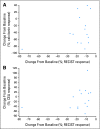
Results: Of 16 patients treated in arm B, one achieved partial response (PR; 6.3%; 95% CI, 0.2% to 30.2%), 14 had stable disease (SD; 87.5%; 95% CI, 61.7% to 99.5%), and one was nonevaluable. In a post hoc analysis of 10 arm B patients with progressive disease (PD) before study, one patient had PR of 21+ months, four patients had SD >or= 15 months, four patients had SD <or= 6 months, and one patient had clinical PD. Median progression-free survival was 17.9 months. Arm A was prematurely terminated because of slow accrual. Common adverse events (AEs) included diarrhea, hand-foot-skin reaction, rash, and hypertension. Although serious AEs were rare, one death was seen. Tumor markers decreased in the majority of patients, and RET mutations were detected in 10 of 12 sporadic MTCs analyzed.
Conclusion: Sorafenib is reasonably well tolerated, with suggestion of clinical benefit for patients with sporadic MTC. Caution should be taken because of the rare but fatal toxicity potentially associated with sorafenib.
Conflict of interest statement
Authors' disclosures of potential conflicts of interest and author contributions are found at the end of this article.
Figures

Comment in
-
Parenchymal versus nonparenchymal target lesion response in clinical trials for metastatic medullary thyroid cancer.J Clin Oncol. 2010 Oct 1;28(28):e534: author reply e535-6. doi: 10.1200/JCO.2010.29.9909. Epub 2010 Aug 16. J Clin Oncol. 2010. PMID: 20713862 No abstract available.
Similar articles
-
Medullary thyroid cancer: targeting the RET kinase pathway with sorafenib/tipifarnib.Mol Cancer Ther. 2008 May;7(5):1001-6. doi: 10.1158/1535-7163.MCT-07-2422. Epub 2008 Apr 29. Mol Cancer Ther. 2008. PMID: 18445656
-
Vandetanib for the treatment of patients with locally advanced or metastatic hereditary medullary thyroid cancer.J Clin Oncol. 2010 Feb 10;28(5):767-72. doi: 10.1200/JCO.2009.23.6604. Epub 2010 Jan 11. J Clin Oncol. 2010. PMID: 20065189 Free PMC article. Clinical Trial.
-
Inhibition of the Ras/Raf/MEK/ERK and RET kinase pathways with the combination of the multikinase inhibitor sorafenib and the farnesyltransferase inhibitor tipifarnib in medullary and differentiated thyroid malignancies.J Clin Endocrinol Metab. 2011 Apr;96(4):997-1005. doi: 10.1210/jc.2010-1899. Epub 2011 Feb 2. J Clin Endocrinol Metab. 2011. PMID: 21289252 Free PMC article. Clinical Trial.
-
Sorafenib: a review of its use in advanced hepatocellular carcinoma.Drugs. 2009;69(2):223-40. doi: 10.2165/00003495-200969020-00006. Drugs. 2009. PMID: 19228077 Review.
-
Preclinical and clinical development of the oral multikinase inhibitor sorafenib in cancer treatment.Drugs Today (Barc). 2005 Dec;41(12):773-84. doi: 10.1358/dot.2005.41.12.937959. Drugs Today (Barc). 2005. PMID: 16474853 Review.
Cited by
-
Sorafenib and Mek inhibition is synergistic in medullary thyroid carcinoma in vitro.Endocr Relat Cancer. 2012 Jan 9;19(1):29-38. doi: 10.1530/ERC-11-0155. Print 2012 Feb. Endocr Relat Cancer. 2012. PMID: 22109971 Free PMC article.
-
Potential therapeutic approaches for modulating expression and accumulation of defective lamin A in laminopathies and age-related diseases.J Mol Med (Berl). 2012 Dec;90(12):1361-89. doi: 10.1007/s00109-012-0962-4. Epub 2012 Oct 23. J Mol Med (Berl). 2012. PMID: 23090008 Free PMC article. Review.
-
Retrospective Study of Sirolimus and Cyclophosphamide in Patients with Advanced Differentiated Thyroid Cancers.J Thyroid Disord Ther. 2015 Aug;4(3):188. doi: 10.4172/2167-7948.1000188. Epub 2015 Jul 17. J Thyroid Disord Ther. 2015. PMID: 27088062 Free PMC article.
-
Activated RET and ROS: two new driver mutations in lung adenocarcinoma.Transl Lung Cancer Res. 2013 Apr;2(2):112-21. doi: 10.3978/j.issn.2218-6751.2013.03.08. Transl Lung Cancer Res. 2013. PMID: 25806222 Free PMC article. Review.
-
Medullary thyroid cancer and pseudocirrhosis: case report and literature review.Curr Oncol. 2012 Feb;19(1):e36-41. doi: 10.3747/co.19.840. Curr Oncol. 2012. PMID: 22328846 Free PMC article.
References
-
- Hoff AO, Hoff PM. Medullary thyroid carcinoma. Hematol Oncol Clin North Am. 2007;21:475–488. - PubMed
-
- Eng C, Clayton D, Schuffenecker I, et al. The relationship between specific RET proto-oncogene mutations and disease phenotype in multiple endocrine neoplasia type 2: International RET mutation consortium analysis. JAMA. 1996;276:1575–1579. - PubMed
-
- Brunt LM, Wells SA., Jr Advances in the diagnosis and treatment of medullary thyroid carcinoma. Surg Clin North Am. 1987;67:263–279. - PubMed
-
- Hong D, Ye L, Gagel R, et al. Medullary thyroid cancer: Targeting the RET kinase pathway with sorafenib/tipifarnib. Mol Cancer Ther. 2008;7:1001–1006. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

