TNFalpha induces choroid plexus epithelial cell barrier alterations by apoptotic and nonapoptotic mechanisms
- PMID: 20369072
- PMCID: PMC2847764
- DOI: 10.1155/2010/307231
TNFalpha induces choroid plexus epithelial cell barrier alterations by apoptotic and nonapoptotic mechanisms
Abstract
The choroid plexus epithelium constitutes the structural basis of the blood-cerebrospinal fluid barrier. Since the cytokine TNFalpha is markedly increased during inflammatory diseases in the blood and the central nervous system, we investigated by which mechanisms TNFalpha induces barrier alteration in porcine choroid plexus epithelial cells. We found a dose-dependent decrease of transepithelial electrical resistance, increase of paracellular inulin-flux, and induction of histone-associated DNA fragmentation and caspase-3 activation after TNFalpha stimulation. This response was strongly aggravated by the addition of cycloheximide and could partially be inhibited by the NF-kappaB inhibitor CAPE, but most effectively by the pan-caspase-inhibitor zVAD-fmk and not by the JNK inhibitor SP600125. Partial loss of cell viability could also be attenuated by CAPE. Immunostaining showed cell condensation and nuclear binding of high-mobility group box 1 protein as a sign of apoptosis after TNFalpha stimulation. Taken together our findings indicate that TNFalpha compromises PCPEC barrier function by caspase and NF-kappaB dependent mechanisms.
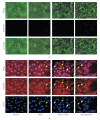
Figures



References
-
- Strazielle N, Ghersi-Egea J-F. Choroid plexus in the central nervous system: biology and physiopathology. Journal of Neuropathology and Experimental Neurology. 2000;59(7):561–574. - PubMed
-
- Engelhardt B, Wolburg-Buchholz K, Wolburg H. Involvement of the choroid plexus in central nervous system inflammation. Microscopy Research and Technique. 2001;52(1):112–129. - PubMed
-
- Balda MS, Matter K. Tight junctions. Journal of Cell Science. 1998;111(5):541–547. - PubMed
-
- Denker BM, Nigam SK. Molecular structure and assembly of the tight junction. American Journal of Physiology. 1998;274(1):F1–F9. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

