Revisiting the loading dose of amikacin for patients with severe sepsis and septic shock
- PMID: 20370907
- PMCID: PMC2887170
- DOI: 10.1186/cc8945
Revisiting the loading dose of amikacin for patients with severe sepsis and septic shock
Abstract
Introduction: It has been proposed that doses of amikacin of >15 mg/kg should be used in conditions associated with an increased volume of distribution (Vd), such as severe sepsis and septic shock. The primary aim of this study was to determine whether 25 mg/kg (total body weight) of amikacin is an adequate loading dose for these patients.
Methods: This was an open, prospective, multicenter study in four Belgian intensive care units (ICUs). All consecutive patients with a diagnosis of severe sepsis or septic shock, in whom amikacin treatment was indicated, were included in the study.
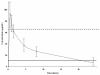
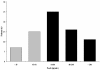
Results: In 74 patients, serum samples were collected before (t = 0 h) and 1 hour (peak), 1 hour 30 minutes, 4 hours 30 minutes, 8 hours, and 24 hours after the first dose of amikacin. Blood amikacin levels were measured by using a validated fluorescence polarization immunoassay method, and an open two-compartment model with first-order elimination was fitted to concentrations-versus-time data for amikacin (WinNonlin). In 52 (70%) patients, peak serum concentrations were >64 microg/ml, which corresponds to 8 times the clinical minimal inhibitory concentration (MIC) breakpoints defined by EUCAST for Enterobacteriaceae and Pseudomonas aeruginosa (S<8, R>16 microg/ml). Vd was 0.41 (0.29 to 0.51) L/kg; elimination half-life, 4.6 (3.2 to 7.8) hours; and total clearance, 1.98 (1.28 to 3.54) ml/min/kg. No correlation was found between the amikacin peak and any clinical or hemodynamic variable.
Conclusions: As patients with severe sepsis and septic shock have an increased Vd, a first dose of >or= 25 mg/kg (total body weight) of amikacin is required to reach therapeutic peak concentrations. However, even with this higher amikacin dose, the peak concentration remained below therapeutic target levels in about one third of these patients. Optimizing aminoglycoside therapy should be achieved by tight serum-concentration monitoring because of the wide interindividual variability of pharmacokinetic abnormalities.
Figures
References
-
- Vincent JL, Taccone F, Schmit X. Classification, incidence, and outcomes of sepsis and multiple organ failure. Contrib Nephrol. 2007;156:64–74. full_text. - PubMed
-
- Bochud PY, Glauser MP, Carlet J, Calandra T. In: The Sepsis Text. Vincent JL, Carlet J, Opal SM, editor. Norwell: Kluwer Academic; 2002. Empirical antibiotic therapy for patients with severe sepsis and septic shock; pp. 539–558. full_text.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

