Thrombin flux and wall shear rate regulate fibrin fiber deposition state during polymerization under flow
- PMID: 20371335
- PMCID: PMC2849060
- DOI: 10.1016/j.bpj.2009.12.4275
Thrombin flux and wall shear rate regulate fibrin fiber deposition state during polymerization under flow
Abstract
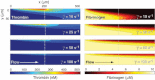
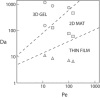
Thrombin is released as a soluble enzyme from the surface of platelets and tissue-factor-bearing cells to trigger fibrin polymerization during thrombosis under flow conditions. Although isotropic fibrin polymerization under static conditions involves protofibril extension and lateral aggregation leading to a gel, factors regulating fiber growth are poorly quantified under hemodynamic flow due to the difficulty of setting thrombin fluxes. A membrane microfluidic device allowed combined control of both thrombin wall flux (10(-13) to 10(-11) nmol/mum(2) s) and the wall shear rate (10-100 s(-1)) of a flowing fibrinogen solution. At a thrombin flux of 10(-12) nmol/mum(2) s, both fibrin deposition and fiber thickness decreased as the wall shear rate increased from 10 to 100 s(-1). Direct measurement and transport-reaction simulations at 12 different thrombin flux-wall shear rate conditions demonstrated that two dimensionless numbers, the Peclet number (Pe) and the Damkohler number (Da), defined a state diagram to predict fibrin morphology. For Da < 10, we only observed thin films at all Pe. For 10 < Da < 900, we observed either mat fibers or gels, depending on the Pe. For Da > 900 and Pe < 100, we observed three-dimensional gels. These results indicate that increases in wall shear rate quench first lateral aggregation and then protofibril extension.
Copyright (c) 2010 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Minimum wound size for clotting: flowing blood coagulates on a single collagen fiber presenting tissue factor and von Willebrand factor.Integr Biol (Camb). 2016 Aug 8;8(8):813-20. doi: 10.1039/c6ib00077k. Epub 2016 Jun 24. Integr Biol (Camb). 2016. PMID: 27339024 Free PMC article.
-
Toward an understanding of fibrin branching structure.Phys Rev E Stat Nonlin Soft Matter Phys. 2010 May;81(5 Pt 1):051922. doi: 10.1103/PhysRevE.81.051922. Epub 2010 May 24. Phys Rev E Stat Nonlin Soft Matter Phys. 2010. PMID: 20866276 Free PMC article.
-
Dynamics of Thrombin Generation and Flux from Clots during Whole Human Blood Flow over Collagen/Tissue Factor Surfaces.J Biol Chem. 2016 Oct 28;291(44):23027-23035. doi: 10.1074/jbc.M116.754671. Epub 2016 Sep 7. J Biol Chem. 2016. PMID: 27605669 Free PMC article.
-
Fibrin gels and their clinical and bioengineering applications.J R Soc Interface. 2009 Jan 6;6(30):1-10. doi: 10.1098/rsif.2008.0327. J R Soc Interface. 2009. PMID: 18801715 Free PMC article. Review.
-
Fibrinogen and fibrin polymerization: appraisal of the binding events that accompany fibrin generation and fibrin clot assembly.Blood Coagul Fibrinolysis. 1997 Jul;8(5):257-67. Blood Coagul Fibrinolysis. 1997. PMID: 9282789 Review.
Cited by
-
The influence of hindered transport on the development of platelet thrombi under flow.Bull Math Biol. 2013 Aug;75(8):1255-83. doi: 10.1007/s11538-012-9784-3. Epub 2012 Oct 25. Bull Math Biol. 2013. PMID: 23097125 Free PMC article.
-
Thrombosis in the pathogenesis of abdominal aortic aneurysm.JVS Vasc Sci. 2023 Apr 14;4:100106. doi: 10.1016/j.jvssci.2023.100106. eCollection 2023. JVS Vasc Sci. 2023. PMID: 37564632 Free PMC article. Review.
-
Data-driven Modeling of Hemodynamics and its Role on Thrombus Size and Shape in Aortic Dissections.Sci Rep. 2018 Feb 6;8(1):2515. doi: 10.1038/s41598-018-20603-x. Sci Rep. 2018. PMID: 29410467 Free PMC article.
-
Flow rate and fibrin fiber alignment.J Thromb Haemost. 2010 Dec;8(12):2826-8. doi: 10.1111/j.1538-7836.2010.04118.x. J Thromb Haemost. 2010. PMID: 20961393 Free PMC article. No abstract available.
-
α-α Cross-links increase fibrin fiber elasticity and stiffness.Biophys J. 2012 Jan 4;102(1):168-75. doi: 10.1016/j.bpj.2011.11.4016. Epub 2012 Jan 3. Biophys J. 2012. PMID: 22225811 Free PMC article.
References
-
- Carr M.E., Powers P.L. Differential effects of divalent cations on fibrin structure. Blood Coagul. Fibrinolysis. 1991;2:741–747. - PubMed
-
- Carr M.E., Jr., Hermans J. Size and density of fibrin fibers from turbidity. Macromolecules. 1978;11:46–50. - PubMed
-
- Hantgan R.R., Hermans J. Assembly of fibrin. A light scattering study. J. Biol. Chem. 1979;254:11272–11281. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

