Modeling an anti-amyloid combination therapy for Alzheimer's disease
- PMID: 20371462
- PMCID: PMC2852193
- DOI: 10.1126/scitranslmed.3000337
Modeling an anti-amyloid combination therapy for Alzheimer's disease
Abstract
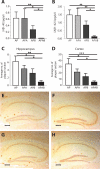
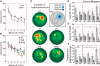
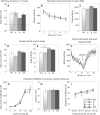
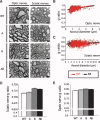
As only symptomatic treatments are now available for Alzheimer's disease (AD), safe and effective mechanism-based therapies remain a great unmet need for patients with this neurodegenerative disease. Although gamma-secretase and BACE1 [beta-site beta-amyloid (Abeta) precursor protein (APP) cleaving enzyme 1] are well-recognized therapeutic targets for AD, untoward side effects associated with strong inhibition or reductions in amounts of these aspartyl proteases have raised concerns regarding their therapeutic potential. Although moderate decreases of either gamma-secretase or BACE1 are not associated with mechanism-based toxicities, they provide only modest benefits in reducing Abeta in the brains of APPswe/PS1DeltaE9 mice. Because the processing of APP to generate Abeta requires both gamma-secretase and BACE1, it is possible that moderate reductions of both enzymes would provide additive and significant protection against Abeta amyloidosis. Here, we test this hypothesis and assess the value of this novel anti-amyloid combination therapy in mutant mice. We demonstrate that genetic reductions of both BACE1 and gamma-secretase additively attenuate the amyloid burden and ameliorate cognitive deficits occurring in aged APPswe/PS1DeltaE9 animals. No evidence of mechanism-based toxicities was associated with such decreases in amounts of both enzymes. Thus, we propose that targeting both gamma-secretase and BACE1 may be an effective and safe treatment strategy for AD.
Figures

Similar articles
-
Amyloid-β protein (Aβ) Glu11 is the major β-secretase site of β-site amyloid-β precursor protein-cleaving enzyme 1(BACE1), and shifting the cleavage site to Aβ Asp1 contributes to Alzheimer pathogenesis.Eur J Neurosci. 2013 Jun;37(12):1962-9. doi: 10.1111/ejn.12235. Eur J Neurosci. 2013. PMID: 23773065
-
Berberine Alleviates Amyloid-Beta Pathology in the Brain of APP/PS1 Transgenic Mice via Inhibiting β/γ-Secretases Activity and Enhancing α-Secretases.Curr Alzheimer Res. 2018;15(11):1045-1052. doi: 10.2174/1567205015666180702105740. Curr Alzheimer Res. 2018. PMID: 29962345
-
BACE1 Cleavage Site Selection Critical for Amyloidogenesis and Alzheimer's Pathogenesis.J Neurosci. 2017 Jul 19;37(29):6915-6925. doi: 10.1523/JNEUROSCI.0340-17.2017. Epub 2017 Jun 16. J Neurosci. 2017. PMID: 28626014 Free PMC article.
-
BACE1: the beta-secretase enzyme in Alzheimer's disease.J Mol Neurosci. 2004;23(1-2):105-14. doi: 10.1385/JMN:23:1-2:105. J Mol Neurosci. 2004. PMID: 15126696 Review.
-
The beta-secretase, BACE: a prime drug target for Alzheimer's disease.J Mol Neurosci. 2001 Oct;17(2):157-70. doi: 10.1385/JMN:17:2:157. J Mol Neurosci. 2001. PMID: 11816789 Review.
Cited by
-
The beta-secretase enzyme BACE in health and Alzheimer's disease: regulation, cell biology, function, and therapeutic potential.J Neurosci. 2009 Oct 14;29(41):12787-94. doi: 10.1523/JNEUROSCI.3657-09.2009. J Neurosci. 2009. PMID: 19828790 Free PMC article. Review.
-
Arc Oligomerization Is Regulated by CaMKII Phosphorylation of the GAG Domain: An Essential Mechanism for Plasticity and Memory Formation.Mol Cell. 2019 Jul 11;75(1):13-25.e5. doi: 10.1016/j.molcel.2019.05.004. Epub 2019 May 28. Mol Cell. 2019. PMID: 31151856 Free PMC article.
-
Alzheimer's disease drug-development pipeline: few candidates, frequent failures.Alzheimers Res Ther. 2014 Jul 3;6(4):37. doi: 10.1186/alzrt269. eCollection 2014. Alzheimers Res Ther. 2014. PMID: 25024750 Free PMC article.
-
BDNF-producing, amyloid β-specific CD4 T cells as targeted drug-delivery vehicles in Alzheimer's disease.EBioMedicine. 2019 May;43:424-434. doi: 10.1016/j.ebiom.2019.04.019. Epub 2019 May 11. EBioMedicine. 2019. PMID: 31085101 Free PMC article.
-
A Review of Pharmacology, Toxicity and Pharmacokinetics of 2,3,5,4'-Tetrahydroxystilbene-2-O-β-D-Glucoside.Front Pharmacol. 2022 Jan 5;12:791214. doi: 10.3389/fphar.2021.791214. eCollection 2021. Front Pharmacol. 2022. PMID: 35069206 Free PMC article. Review.
References
-
- Hardy J, Selkoe DJ. The amyloid hypothesis of Alzheimer's disease: Progress and problems on the road to therapeutics. Science. 2002;297:353–356. - PubMed
-
- Tanzi RE, Bertram L. Twenty years of the Alzheimer's disease amyloid hypothesis: A genetic perspective. Cell. 2005;120:545–555. - PubMed
-
- Wong PC, Cai H, Borchelt DR, Price DL. Genetically engineered mouse models of neurodegenerative diseases. Nat. Neurosci. 2002;5:633–639. - PubMed
-
- De Strooper B. Aph-1, Pen-2, and Nicastrin with Presenilin generate an active γ-Secretase complex. Neuron. 2003;38:9–12. - PubMed
-
- Li T, Wen H, Brayton C, Das P, Smithson LA, Fauq A, Fan X, Crain BJ, Price DL, Golde TE, Eberhart CG, Wong PC. Epidermal growth factor receptor and Notch pathways participate in the tumor suppressor function of γ-secretase. J. Biol. Chem. 2007;282:32264–32273. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

