MicroRNA-155 regulates cell survival, growth, and chemosensitivity by targeting FOXO3a in breast cancer
- PMID: 20371610
- PMCID: PMC2878550
- DOI: 10.1074/jbc.M110.101055
MicroRNA-155 regulates cell survival, growth, and chemosensitivity by targeting FOXO3a in breast cancer
Retraction in
-
MicroRNA-155 regulates cell survival, growth, and chemosensitivity by targeting FOXO3a in breast cancer.J Biol Chem. 2016 Oct 21;291(43):22855. doi: 10.1074/jbc.A110.101055. J Biol Chem. 2016. PMID: 27825093 Free PMC article. No abstract available.
Abstract
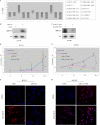
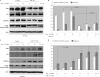
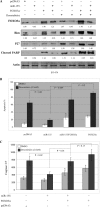
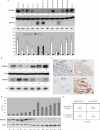
Breast cancer is the second leading cause of cancer death in women. Despite improvement in treatment over the past few decades, there is an urgent need for development of targeted therapies. miR-155 (microRNA-155) is frequently up-regulated in breast cancer. In this study, we demonstrate the critical role of miR-155 in regulation of cell survival and chemosensitivity through down-regulation of FOXO3a in breast cancer. Ectopic expression of miR-155 induces cell survival and chemoresistance to multiple agents, whereas knockdown of miR-155 renders cells to apoptosis and enhances chemosensitivity. Further, we identified FOXO3a as a direct target of miR-155. Sustained overexpression of miR-155 resulted in repression of FOXO3a protein without changing mRNA levels, and knockdown of miR-155 increases FOXO3a. Introduction of FOXO3a cDNA lacking the 3'-untranslated region abrogates miR-155-induced cell survival and chemoresistance. Finally, inverse correlation between miR-155 and FOXO3a levels were observed in a panel of breast cancer cell lines and tumors. In conclusion, our study reveals a molecular link between miR-155 and FOXO3a and presents evidence that miR-155 is a critical therapeutic target in breast cancer.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

