Foxo3a regulates apoptosis by negatively targeting miR-21
- PMID: 20371612
- PMCID: PMC2878079
- DOI: 10.1074/jbc.M109.093005
Foxo3a regulates apoptosis by negatively targeting miR-21
Abstract
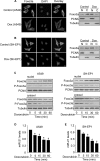
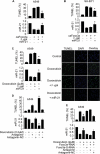
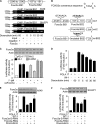
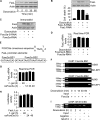
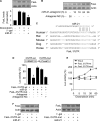
MicroRNAs are a class of small non-coding RNAs and participate in the regulation of apoptotic program. Although miR-21 is able to inhibit apoptosis, its expression regulation and downstream targets remain to be fully elucidated. Here we report that the transcriptional factor Foxo3a initiates apoptosis by transcriptionally repressing miR-21 expression. Our results showed that doxorubicin could simultaneously induce the translocation of Foxo3a to the cell nuclei and a reduction in miR-21 expression. Knockdown of Foxo3a resulted in an elevation in miR-21 levels, whereas enforced expression of Foxo3a led to a decrease in miR-21 expression. In exploring the molecular mechanism by which Foxo3a regulates miR-21, we observed that Foxo3a bound to the promoter region of miR-21 and suppressed its promoter activity. These results indicate that Foxo3a can transcriptionally repress miR-21 expression. In searching for the downstream targets of miR-21 in apoptosis, we found that miR-21 suppressed the translation of Fas ligand (FasL), a pro-apoptotic factor. Furthermore, Foxo3a was able to up-regulate FasL expression through down-regulating miR-21. Our data suggest that Foxo3a negatively regulates miR-21 in initiating apoptosis.
Figures

Similar articles
-
MicroRNA-155 regulates cell survival, growth, and chemosensitivity by targeting FOXO3a in breast cancer.J Biol Chem. 2010 Jun 4;285(23):17869-79. doi: 10.1074/jbc.M110.101055. Epub 2010 Apr 6. J Biol Chem. 2010. Retraction in: J Biol Chem. 2016 Oct 21;291(43):22855. doi: 10.1074/jbc.A110.101055. PMID: 20371610 Free PMC article. Retracted.
-
miR-874 regulates myocardial necrosis by targeting caspase-8.Cell Death Dis. 2013 Jul 4;4(7):e709. doi: 10.1038/cddis.2013.233. Cell Death Dis. 2013. PMID: 23828572 Free PMC article.
-
MicroRNA-96 promotes the proliferation of colorectal cancer cells and targets tumor protein p53 inducible nuclear protein 1, forkhead box protein O1 (FOXO1) and FOXO3a.Mol Med Rep. 2015 Feb;11(2):1200-6. doi: 10.3892/mmr.2014.2854. Epub 2014 Nov 4. Mol Med Rep. 2015. PMID: 25369914
-
Effects of miR-155 on proliferation and apoptosis by regulating FoxO3a/BIM in liver cancer cell line HCCLM3.Eur Rev Med Pharmacol Sci. 2018 Mar;22(5):1277-1285. doi: 10.26355/eurrev_201803_14468. Eur Rev Med Pharmacol Sci. 2018. Retraction in: Eur Rev Med Pharmacol Sci. 2020 Jul;24(13):7196. doi: 10.26355/eurrev_202007_21845. PMID: 29565484 Retracted.
-
Progress in the study of FOXO3a interacting with microRNA to regulate tumourigenesis development.Front Oncol. 2023 Oct 27;13:1293968. doi: 10.3389/fonc.2023.1293968. eCollection 2023. Front Oncol. 2023. PMID: 37965449 Free PMC article. Review.
Cited by
-
AKT kinase pathway: a leading target in cancer research.ScientificWorldJournal. 2013 Nov 13;2013:756134. doi: 10.1155/2013/756134. ScientificWorldJournal. 2013. PMID: 24327805 Free PMC article. Review.
-
MicroRNA-21 regulates non-small cell lung cancer cell proliferation by affecting cell apoptosis via COX-19.Int J Clin Exp Med. 2015 Jun 15;8(6):8835-41. eCollection 2015. Int J Clin Exp Med. 2015. PMID: 26309536 Free PMC article.
-
FOXOs: signalling integrators for homeostasis maintenance.Nat Rev Mol Cell Biol. 2013 Feb;14(2):83-97. doi: 10.1038/nrm3507. Epub 2013 Jan 17. Nat Rev Mol Cell Biol. 2013. PMID: 23325358 Review.
-
The oncogenic microRNA OncomiR-21 overexpressed during Marek's disease lymphomagenesis is transactivated by the viral oncoprotein Meq.J Virol. 2013 Jan;87(1):80-93. doi: 10.1128/JVI.02449-12. Epub 2012 Oct 10. J Virol. 2013. PMID: 23055556 Free PMC article.
-
Cardioprotective microRNAs.Pediatr Cardiol. 2011 Mar;32(3):311-6. doi: 10.1007/s00246-010-9882-7. Epub 2011 Jan 19. Pediatr Cardiol. 2011. PMID: 21243487
References
-
- Park S. M., Schickel R., Peter M. E. (2005) Curr. Opin. Cell Biol. 17, 610–616 - PubMed
-
- Löffler D., Brocke-Heidrich K., Pfeifer G., Stocsits C., Hackermüller J., Kretzschmar A. K., Burger R., Gramatzki M., Blumert C., Bauer K., Cvijic H., Ullmann A. K., Stadler P. F., Horn F. (2007) Blood 110, 1330–1333 - PubMed
-
- Accili D., Arden K. C. (2004) Cell 117, 421–426 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

